Scrub typhus
Last updated| Scrub typhus | |
|---|---|
| Other names | Bush typhus, mite typhus, jungle typhus [1] |
 | |
| Orientia tsutsugamushi | |
| Specialty | Infectious disease |
Scrub typhus or bush typhus is a form of typhus caused by the intracellular parasite Orientia tsutsugamushi , a Gram-negative α-proteobacterium of family Rickettsiaceae first isolated and identified in 1930 in Japan. [2] [3]
Contents
Although the disease is similar in presentation to other forms of typhus, its pathogen is no longer included in genus Rickettsia with the typhus bacteria proper, but in Orientia . The disease is thus frequently classified separately from the other typhi.
Signs and symptoms
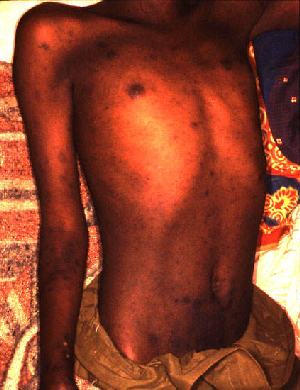
Signs and symptoms include fever, headache, muscle pain, cough, and gastrointestinal symptoms. More virulent strains of O. tsutsugamushi can cause hemorrhaging and intravascular coagulation. Morbilliform rash, eschar, splenomegaly, and lymphadenopathies are typical signs. Leukopenia and abnormal liver function tests are commonly seen in the early phase of the illness. Pneumonitis, encephalitis, and myocarditis occur in the late phase of illness. It has particularly been shown to be the most common cause of acute encephalitis syndrome in Bihar, India. [4]
Causes
Scrub typhus is transmitted by some species of trombiculid mites ("chiggers", particularly Leptotrombidium deliense ), [5] which are found in areas of heavy scrub vegetation. The mites feed on infected rodent hosts and subsequently transmit the parasite to other rodents and humans. The bite of this mite leaves a characteristic black eschar that is useful to the doctor for making the diagnosis. [6]
Scrub typhus is endemic to a part of the world known as the tsutsugamushi triangle (after O. tsutsugamushi ). [3] This extends from northern Japan and far-eastern Russia in the north, to the territories around the Solomon Sea into northern Australia in the south, and to Pakistan and Afghanistan in the west. [7] It may also be endemic in parts of South America. [8]
The precise incidence of the disease is unknown, as diagnostic facilities are not available in much of its large native range, which spans vast regions of equatorial jungle to the subtropics. In rural Thailand and Laos, murine and scrub typhus account for around a quarter of all adults presenting to hospital with fever and negative blood cultures. [9] [10] The incidence in Japan has fallen over the past few decades, probably due to land development driving decreasing exposure, and many prefectures report fewer than 50 cases per year. [11] [12]
It affects females more than males in Korea, but not in Japan, [13] which may be because sex-differentiated cultural roles have women tending garden plots more often, thus being exposed to vegetation inhabited by chiggers. The incidence is increasing in the southern part of the Indian subcontinent and in northern areas around Darjeeling.[ citation needed ]
Diagnosis
In endemic areas, diagnosis is generally made on clinical grounds alone. However, overshadowing of the diagnosis is quite often as the clinical symptoms overlap with other infectious diseases such as dengue fever, paratyphoid, and pyrexia of unknown origin (PUO). If the eschar can be identified, it is quite diagnostic of scrub typhus, but this can be unreliable on dark skin, and moreover, the site of eschar which is usually where the mite bites is often located in covered areas. Unless it is actively searched for, the eschar can easily be missed. History of mite bite is often absent since the bite does not inflict pain and the mites are almost too small to be seen by the naked eye. Usually, scrub typhus is often labelled as PUO in remote endemic areas, since blood culture is often negative, yet it can be treated effectively with chloramphenicol. Where doubt exists, the diagnosis may be confirmed by a laboratory test such as serology. Again, this is often unavailable in most endemic areas, since the serological test involved is not included in the routine screening tests for PUO, especially in Burma (Myanmar).[ citation needed ]
The choice of laboratory test is not straightforward, and all currently available tests have their limitations. [14] The cheapest and most easily available serological test is the Weil-Felix test, but this is notoriously unreliable. [15] The best test is indirect immunofluorescence, [16] but the main limitation of this method is the availability of fluorescent microscopes, which are not often available in resource-poor settings where scrub typhus is endemic. Indirect immunoperoxidase, a modification of the standard IFA method, can be used with a light microscope, [17] and the results of these tests are comparable to those from IFA. [15] [18] Rapid bedside kits have been described that produce a result within one hour, but the availability of these tests is severely limited by their cost. [15] Serological methods are most reliable when a four-fold rise in antibody titre is found. If the patient is from a nonendemic area, then diagnosis can be made from a single acute serum sample. [19] In patients from endemic areas, this is not possible because antibodies may be found in up to 18% of healthy individuals. [20]
Other methods include culture and polymerase chain reaction, but these are not routinely available [21] and the results do not always correlate with serological testing, [22] [23] [24] and are affected by prior antibiotic treatment. [25] The currently available diagnostic methods have been summarised. [14]
Treatment
Without treatment, the disease is often fatal. Since the use of antibiotics, case fatalities have decreased from 4–40% to less than 2%.[ citation needed ]
The drug most commonly used is doxycycline or tetracycline, but chloramphenicol is an alternative. Strains that are resistant to doxycycline and chloramphenicol have been reported in northern Thailand. [26] [27] Rifampicin [28] and azithromycin [29] are alternatives. Azithromycin is an alternative in children [30] and pregnant women with scrub typhus, [31] [32] [33] and when doxycycline resistance is suspected. [34] Ciprofloxacin cannot be used safely in pregnancy and is associated with stillbirths and miscarriage. [33] [35] Combination therapy with doxycycline and rifampicin is not recommended due to possible antagonism. [36]
Vaccine
No licensed vaccines are available. [37]
An early attempt to create a scrub typhus vaccine occurred in the United Kingdom in 1937 (with the Wellcome Foundation infecting around 300,000 cotton rats in a classified project called "Operation Tyburn"), but the vaccine was not used. [38] The first known batch of scrub typhus vaccine actually used to inoculate human subjects was dispatched to India for use by Allied Land Forces, South-East Asia Command in June 1945. By December 1945, 268,000 cc had been dispatched. [39] The vaccine was produced at Wellcome's laboratory at Ely Grange, Frant, Sussex. An attempt to verify the efficacy of the vaccine by using a placebo group for comparison was vetoed by the military commanders, who objected to the experiment. [40]
Enormous antigenic variation in Orientia tsutsugamushi strains is now recognized, [41] [42] and immunity to one strain does not confer immunity to another. Any scrub typhus vaccine should give protection to all the strains present locally, to give an acceptable level of protection. A vaccine developed for one locality may not be protective in another, because of antigenic variation. This complexity continues to hamper efforts to produce a viable vaccine. [43]
Dora Lush, an Australian bacteriologist, died after accidentally pricking her finger with a needle containing scrub typhus while inoculating a mouse in an attempt to develop a vaccine. [44]
History

Severe epidemics of the disease occurred among troops in Burma and Ceylon during World War II. [45] Several members of the U.S. Army's 5307th Composite Unit (Merrill's Marauders) died of the disease, as well as many soldiers in the Burma theatre; [46] and before 1944, no effective antibiotics or vaccines were available. [47] [48]
World War II provides some indicators that the disease is endemic to undeveloped areas in all of Oceania in the Pacific theater, although war records frequently lack definitive diagnoses, and many records of "high fever" evacuations were also likely to be other tropical illnesses. In the chapter entitled "The Green War", General MacArthur's biographer William Manchester identifies that the disease was one of a number of debilitating afflictions affecting both sides on New Guinea [49] in the running bloody Kokoda battles over extremely harsh terrains under intense hardships— fought during a six-month span [50] all along the Kokoda Track in 1942–43, and mentions that to be hospital-evacuated, Allied soldiers (who cycled forces) had to run a fever of 102 °F (39 °C), and that sickness casualties outnumbered weapons-inflicted casualties 5:1. [49] Similarly, the illness was a casualty producer in all the jungle fighting of the land battles of the New Guinea campaign and the Guadalcanal campaign. Where the Allies had bases, they could remove and cut back vegetation, or use DDT as a prophylaxis area barrier treatment, so mite- and tick-induced sickness rates in forces off the front lines were diminished.[ citation needed ]
The disease was also a problem for US troops stationed in Japan after WWII, and was variously known as "Shichitō fever" (by troops stationed in the Izu Seven Islands) or "Hatsuka fever" (Chiba prefecture). [51]
Scrub typhus was first reported in Chile in 2006. [52] This is likely the result of underdiagnosis and underreporting and not of a recent spread to Chile. [52] In January 2020 the disease was for the first time reported in Chile's southernmost region. [52]
See also
Related Research Articles

Boutonneuse fever is a fever as a result of a rickettsial infection caused by the bacterium Rickettsia conorii and transmitted by the dog tick Rhipicephalus sanguineus. Boutonneuse fever can be seen in many places around the world, although it is endemic in countries surrounding the Mediterranean Sea. This disease was first described in Tunisia in 1910 by Conor and Bruch and was named boutonneuse due to its papular skin-rash characteristics.
Bolivian hemorrhagic fever (BHF), also known as black typhus or Ordog Fever, is a hemorrhagic fever and zoonotic infectious disease originating in Bolivia after infection by Machupo mammarenavirus.

Leptospirosis is a blood infection caused by the bacteria Leptospira that can infect humans, dogs, rodents and many other wild and domesticated animals. Signs and symptoms can range from none to mild to severe. Weil's disease, the acute, severe form of leptospirosis, causes the infected individual to become jaundiced, develop kidney failure, and bleed. Bleeding from the lungs associated with leptospirosis is known as severe pulmonary haemorrhage syndrome.
The Weil–Felix test is an agglutination test for the diagnosis of rickettsial infections. It was first described in 1916. By virtue of its long history and of its simplicity, it has been one of the most widely employed tests for rickettsia on a global scale, despite being superseded in many settings by more sensitive and specific diagnostic tests. The Weil-Felix antibody was recently found to target rickettsia LPS O-antigen.
Relapsing fever is a vector-borne disease caused by infection with certain bacteria in the genus Borrelia, which is transmitted through the bites of lice or soft-bodied ticks.

Orientia is a genus of bacteria in family Rickettsiaceae. They are obligate intracellular, gram-negative bacteria found in insects and mammals. They are spread through the bites or feces of infected insects.

Ehrlichiosis is a tick-borne bacterial infection, caused by bacteria of the family Anaplasmataceae, genera Ehrlichia and Anaplasma. These obligate intracellular bacteria infect and kill white blood cells.
A rickettsiosis is a disease caused by intracellular bacteria.

Bartonella bacilliformis is a bacterium, Gram negative aerobic, pleomorphic, flagellated, motile, coccobacillary, 2–3 μm long, 0.2–0.5 μm wide, and a facultative intracellular bacterium.
Bartonella quintana, originally known as Rochalimaea quintana, and "Rickettsia quintana", is a bacterium transmitted by the human body louse that causes trench fever. This bacterial species caused outbreaks of trench fever affecting 1 million soldiers in Europe during World War I.

Orientia tsutsugamushi is a mite-borne bacterium belonging to the family Rickettsiaceae and is responsible for a disease called scrub typhus in humans. It is a natural and an obligate intracellular parasite of mites belonging to the family Trombiculidae. With a genome of only 2.0–2.7 Mb, it has the most repeated DNA sequences among bacterial genomes sequenced so far. The disease, scrub typhus, occurs when infected mite larvae accidentally bite humans. Primarily indicated by undifferentiated febrile illnesses, the infection can be complicated and often fatal.
Typhus, also known as typhus fever, is a group of infectious diseases that include epidemic typhus, scrub typhus, and murine typhus. Common symptoms include fever, headache, and a rash. Typically these begin one to two weeks after exposure.
Leptotrombidium is a genus of mites in the family Trombiculidae, that are able to infect humans with scrub typhus through their bite. The larval form feeds on rodents, but also occasionally humans and other large mammals. They are related to the harvest mites of the North America and Europe.

Deer tick virus (DTV) is a virus in the genus Flavivirus spread via ticks that causes encephalitis.

African tick bite fever (ATBF) is a bacterial infection spread by the bite of a tick. Symptoms may include fever, headache, muscle pain, and a rash. At the site of the bite there is typically a red skin sore with a dark center. The onset of symptoms usually occurs 4–10 days after the bite. Complications are rare but may include joint inflammation. Some people do not develop symptoms.
Queensland tick typhus is a zoonotic disease caused by the bacterium Rickettsia australis. It is transmitted by the ticks Ixodes holocyclus and Ixodes tasmani.

Rickettsia parkeri is a gram-negative intracellular bacterium. The organism is found in the Western Hemisphere and is transmitted via the bite of hard ticks of the genus Amblyomma. R. parkeri causes mild spotted fever disease in humans, whose most common signs and symptoms are fever, an eschar at the site of tick attachment, rash, headache, and muscle aches. Doxycycline is the most common drug used to reduce the symptoms associated with disease.

The Mazzotti reaction, first described in 1948, is a symptom complex seen in patients after undergoing treatment of nematode infestation, particularly with the medication diethylcarbamazine (DEC). Mazzotti reactions can be life-threatening, and are characterized by fever, urticaria, swollen and tender lymph nodes, tachycardia, hypotension, arthralgias, oedema, and abdominal pain that occur within seven days of treatment of microfilariasis. The Mazzotti reaction correlates with intensity of infection; however, there are probably multiple infection intensity-dependent mechanisms responsible for mediating this complex reaction.
Cryptosporidium muris is a species of coccidium, first isolated from the gastric glands of the common mouse. Cryptosporidium does originate in common mice, specifically laboratory mice. However, it also has infected cows, dogs, cats, rats, rabbits, lambs, and humans and other primates.
Tensaw orthobunyavirus is a virus in the genus Orthobunyavirus of the Bunyamwera arbovirus group, order Bunyavirales. It is named for the river bordering the area in south Alabama where the prototype strain was discovered. It is abbreviated TEN, TENV, and TSV in the scientific literature.
References
- ↑ Slim, William (1956). Defeat Into Victory. London: Sassell. p. 177.
- ↑ Tseng BY, Yang HH, Liou JH, Chen LK, Hsu YH (February 2008). "Immunohistochemical study of scrub typhus: a report of two cases". Kaohsiung J. Med. Sci. 24 (2): 92–8. doi:10.1016/S1607-551X(08)70103-7. PMID 18281226. S2CID 206172576.
- 1 2 Pediatric Scrub Typhus, accessdate: 16 October 2011
- ↑ Jain P, Prakash S, Tripathi PK, Chauhan A, Gupta S, Sharma U (2018). "Emergence of Orientia tsutsugamushi as an important cause of acute encephalitis syndrome in India". PLOS Negl Trop Dis. 12 (3): e0006346. doi: 10.1371/journal.pntd.0006346 . PMC 5891077 . PMID 29590177.
- ↑ Pham XD, Otsuka Y, Suzuki H, Takaoka H (2001). "Detection of Orientia tsutsugamushi (Rickettsiales: Rickettsiaceae) in unengorged chiggers (Acari: Trombiculidae) from Oita Prefecture, Japan, by nested polymerase chain reaction". J Med Entomol. 38 (2): 308–311. doi:10.1603/0022-2585-38.2.308. PMID 11296840. S2CID 8133110.
- ↑ Park J (2016). "Evolution of Eschar in Scrub Typhus". American Journal of Tropical Medicine and Hygiene. 95 (6): 1223–1224. doi:10.4269/ajtmh.16-0583. PMC 5154431 . PMID 27928073.
- ↑ Seong SY, Choi MS, Kim IS (January 2001). "Orientia tsutsugamushi infection: overview and immune responses". Microbes Infect. 3 (1): 11–21. doi: 10.1016/S1286-4579(00)01352-6 . PMID 11226850.
- ↑ Deadly scrub typhus bacteria confirmed in South America. ScienMag (September 8, 2016)
- ↑ Phongmany S, Rolain JM, Phetsouvanh R, Blacksell SD, Soukkhaseum V, Rasachack B, Phiasakha K, Soukkhaseum S, Frichithavong K (February 2006). "Rickettsial infections and fever, Vientiane, Laos". Emerging Infect. Dis. 12 (2): 256–62. doi:10.3201/eid1202.050900. PMC 3373100 . PMID 16494751.
- ↑ Suttinont C, Losuwanaluk K, Niwatayakul K, Hoontrakul S, Intaranongpai W, Silpasakorn S, Suwancharoen D, Panlar P, Saisongkorh W (June 2006). "Causes of acute, undifferentiated, febrile illness in rural Thailand: results of a prospective observational study". Ann Trop Med Parasitol. 100 (4): 363–70. doi:10.1179/136485906X112158. PMID 16762116. S2CID 25778287.
- ↑ Katayama T, Hara M, Furuya Y, Nikkawa T, Ogasawara H (June 2006). "Scrub typhus (tsutsugamushi disease) in Kanagawa Prefecture in 2001–2005". Jpn J Infect Dis. 59 (3): 207–8. PMID 16785710. Archived from the original on 2010-05-25.
- ↑ Yamamoto S, Ganmyo H, Iwakiri A, Suzuki S (December 2006). "Annual incidence of tsutsugamushi disease in Miyazaki prefecture, Japan in 2001-2005". Jpn J Infect Dis. 59 (6): 404–5. PMID 17186964. Archived from the original on 2009-08-15.
- ↑ Bang HA, Lee MJ, Lee WC (2008). "Comparative research on epidemiological aspects of tsutsugamushi disease (scrub typhus) between Korea and Japan". Jpn J Infect Dis. 61 (2): 148–50. doi:10.7883/yoken.JJID.2008.148. PMID 18362409. Archived from the original on 2008-09-27.
- 1 2 Koh GC, Maude RJ, Paris DH, Newton PN, Blacksell SD (March 2010). "Diagnosis of scrub typhus". Am. J. Trop. Med. Hyg. 82 (3): 368–70. doi:10.4269/ajtmh.2010.09-0233. PMC 2829893 . PMID 20207857.
- 1 2 3 Pradutkanchana J, Silpapojakul K, Paxton H, Pradutkanchana S, Kelly DJ, Strickman D (1997). "Comparative evaluation of four serodiagnostic tests for scrub typhus in Thailand". Trans R Soc Trop Med Hyg. 91 (4): 425–8. doi:10.1016/S0035-9203(97)90266-2. PMID 9373640.
- ↑ Bozeman FM & Elisberg BL (1963). "Serological diagnosis of scrub typhus by indirect immunofluorescence". Proc Soc Exp Biol Med. 112 (3): 568–73. doi:10.3181/00379727-112-28107. PMID 14014756. S2CID 44692835.
- ↑ Yamamoto S & Minamishima Y (1982). "Serodiagnosis of tsutsugamushi fever (scrub typhus) by the indirect immunoperoxidase technique". J Clin Microbiol. 15 (6): 1128–l. doi:10.1128/JCM.15.6.1128-1132.1982. PMC 272264 . PMID 6809786.
- ↑ Kelly DJ, Wong PW, Gan E, Lewis GE Jr (1988). "Comparative evaluation of the indirect immunoperoxidase test for the serodiagnosis of rickettsial disease". Am J Trop Med Hyg. 38 (2): 400–6. doi:10.4269/ajtmh.1988.38.400. PMID 3128129.
- ↑ Blacksell SD, Bryant NJ, Paris, DH, Sakoda Y, Day NP (2007). "Scrub typhus serologic testing with the indirect immunofluorescence method as a diagnostic gold standard: a lack of consensus leads to a lot of confusion". Clin Infect Dis. 44 (3): 391–401. doi: 10.1086/510585 . PMID 17205447.
- ↑ Eamsila C, Singsawat P, Duangvaraporn A, Strickman D (1996). "Antibodies to Orientia tsutsugamushi in Thai soldiers". Am J Trop Med Hyg. 55 (5): 556–9. doi:10.4269/ajtmh.1996.55.556. PMID 8940989.
- ↑ Watt G, Parola P (2003). "Scrub typhus and tropical rickettsioses". Curr Opin Infect Dis. 16 (5): 429–436. doi:10.1097/00001432-200310000-00009. PMID 14501995. S2CID 24087729.
- ↑ Tay ST, Nazma S, Rohani MY (1996). "Diagnosis of scrub typhus in Malaysian aborigines using nested polymerase chain reaction". Southeast Asian J Trop Med Public Health. 27 (3): 580–3. PMID 9185274.
- ↑ Kim DM, Yun NR, Yang TY, Lee JH, Yang JT, Shim SK, Choi EN, Park MY, Lee SH (2006). "Usefulness of nested PCR for the diagnosis of scrub typhus in clinical practice: A prospective study". Am J Trop Med Hyg. 75 (3): 542–545. doi: 10.4269/ajtmh.2006.75.542 . PMID 16968938.
- ↑ Sonthayanon P, Chierakul W, Wuthiekanun V, Blacksell SD, Pimda K, Suputtamongkol Y, Pukrittayakamee S, White NJ, Day NP (December 2006). "Rapid diagnosis of scrub typhus in rural Thailand using polymerase chain reaction". Am. J. Trop. Med. Hyg. 75 (6): 1099–102. doi: 10.4269/ajtmh.2006.75.1099 . PMID 17172374.
- ↑ Kim DM, Byun JN (2008). "Effects of Antibiotic Treatment on the Results of Nested PCRs for Scrub Typhus". J Clin Microbiol. 46 (10): 3465–. doi:10.1128/JCM.00634-08. PMC 2566087 . PMID 18716229.
- ↑ Watt G, Chouriyagune C, Ruangweerayud R, Watcharapichat P, Phulsuksombati D, Jongsakul K, Teja-Isavadharm P, Bhodhidatta D, Corcoran KD (1996). "Scrub typhus infections poorly responsive to antibiotics in northern Thailand". Lancet. 348 (9020): 86–89. doi:10.1016/S0140-6736(96)02501-9. PMID 8676722. S2CID 23316592.
- ↑ Kollars TM, Bodhidatta D, Phulsuksombati D, Tippayachai B, Coleman RE (2003). "Short report: variation in the 56-kD type-specific antigen gene of Orientia tsutsugamushi isolated from patients in Thailand". Am J Trop Med Hyg. 68 (3): 299–300. doi: 10.4269/ajtmh.2003.68.299 . PMID 12685633.
- ↑ El Sayed I, Liu Q, Wee I, Hine P (24 September 2018). "Antibiotics for treating scrub typhus". The Cochrane Database of Systematic Reviews. 2018 (9): CD002150. doi:10.1002/14651858.CD002150.pub2. ISSN 1469-493X. PMC 6485465 . PMID 30246875.
- ↑ Phimda K, Hoontrakul S, Suttinont C, Chareonwat S, Losuwanaluk K, Chueasuwanchai S, Chierakul W, Suwancharoen D, Silpasakorn S (2007). "Doxycycline versus Azithromycin for Treatment of Leptospirosis and Scrub Typhus". Antimicrob Agents Chemother. 51 (9): 3259–63. doi:10.1128/AAC.00508-07. PMC 2043199 . PMID 17638700.
- ↑ Mahajan SK, Rolain JM, Sankhyan N, Kaushal RK, Raoult D (2008). "Pediatric scrub typhus in Indian Himalayas". Indian Journal of Pediatrics. 75 (9): 947–9. doi:10.1007/s12098-008-0198-z. PMID 19011809. S2CID 207384950.
- ↑ Watt G, Kantipong P, Jongsakul K, Watcharapichat P, Phulsuksombati D (1999). "Azithromycin Activities against Orientia tsutsugamushi Strains Isolated in Cases of Scrub Typhus in Northern Thailand". Antimicrob Agents Chemother. 43 (11): 2817–2818. doi:10.1128/AAC.43.11.2817. PMC 89570 . PMID 10543774.
- ↑ Choi EK, Pai H (1998). "Azithromycin therapy for scrub typhus during pregnancy". Clin Infect Dis. 27 (6): 1538–9. doi: 10.1086/517742 . PMID 9868680.
- 1 2 Kim YS, Lee HJ, Chang M, Son SK, Rhee YE, Shim SK (2006). "Scrub typhus during pregnancy and its treatment: a case series and review of the literature". Am J Trop Med Hyg. 75 (5): 955–9. doi: 10.4269/ajtmh.2006.75.955 . PMID 17123995.
- ↑ <Please add first missing authors to populate metadata.> (2003). "Efficacy of azithromycin for treatment of mild scrub-typhus infections in South Korea". Abstr Intersci Conf Antimicrob Agents Chemother. 43: abstract no. L–182.
- ↑ Mathai E, Rolain JM, Verghese L, Mathai M, Jasper P, Verghese G, Raoult D (2003). "Case reports: scrub typhus during pregnancy in India". Trans R Soc Trop Med Hyg. 97 (5): 570–2. doi:10.1016/S0035-9203(03)80032-9. PMID 15307429.
- ↑ Watt G, Kantipong P, Jongsakul K, Watcharapichat P, Phulsuksombati D, Strickman D (2000). "Doxycycline and rifampicin for mild scrub-typhus infections in northern Thailand: a randomised trial". Lancet. 356 (9235): 1057–1061. doi:10.1016/S0140-6736(00)02728-8. PMID 11009140. S2CID 29646085.
- ↑ Arguin PM, Kozarsky PE, Reed C (2008). "Chapter 4: Rickettsial Infections". CDC Health Information for International Travel, 2008. Mosby. ISBN 978-0-323-04885-9. Archived from the original on 2009-05-06. Retrieved 2008-10-03.
- ↑ "AWIC Newsletter: The Cotton Rat In Biomedical Research". Archived from the original on 2004-06-10.
- ↑ "Scrub Typhus Vaccine, Far East". Parliamentary Debates (Hansard) . Millbanksystems. April 2, 1946.
- ↑ Thomson Walker W (1947). "Scrub Typhus Vaccine". Br Med J. 1 (4501): 484–7. doi:10.1136/bmj.1.4501.484. PMC 2053023 . PMID 20248030.
- ↑ Shirai A, Tanskul PL, Andre, RG, Huxsoll DL (1981). "Rickettsia tsutsugamushi strains found in chiggers collected in Thailand". Southeast Asian J Trop Med Public Health. 12 (1): 1–6. PMID 6789455.
- ↑ Kang JS, Chang WH (1999). "Antigenic relationship among the eight prototype and new serotype strains of Orientia tsutsugamushi revealed by monoclonal antibodies". Microbiol Immunol. 43 (3): 229–34. doi: 10.1111/j.1348-0421.1999.tb02397.x . PMID 10338191. S2CID 41165633.
- ↑ Kelly DJ, Fuerst PA, Ching WM, Richards AL (2009). "Scrub typhus: The geographic distribution of phenotypic and genotypic variants of Orientia tsutsugamushi". Clinical Infectious Diseases. 48 (s3): S203–30. doi:10.1086/596576. PMID 19220144.
- ↑ "Death from Scrub Fever". Kalgoorlie Miner. 1943-05-25. Retrieved 2022-06-21.
- ↑ Audy JR (1968). Red mites and typhus. London: University of London, Athlone Press. ISBN 978-0-485-26318-3.
- ↑ Grant, Ian Lyall- (2003). Burma: The Turning Point. Barnsley, South Yorkshire: Leo Cooper. p. 212. ISBN 1-84415-026-7.
- ↑ Kearny CH (1997). Jungle Snafus...And Remedies. Cave Junction, Oregon: Oregon Institute of Science & Medicine. p. 309. ISBN 978-1-884067-10-5.
- ↑ Smallman-Raynor M, Cliff AD (2004). War epidemics: an historical geography of infectious diseases in military conflict and civil strife, 1850–2000. Oxford: Oxford University Press. pp. 489–91. ISBN 978-0-19-823364-0.
- 1 2 William Manchester (1978). "The Green War". American Caesar. Little Brown Company. pp. 297–298. ISBN 978-0-316-54498-6.
- ↑ Manchester, p. Six months to recapture Buna and Gona from July 21–22, 1942
- ↑ Ogawa M, Hagiwara T, Kishimoto T, Shiga S, Yoshida Y, Furuya Y, Kaiho I, Ito T, Nemoto H (1 August 2002). "Scrub typhus in Japan: Epidemiology and clinical features of cases reported in 1998". Am J Trop Med Hyg. 67 (2): 162–5. doi: 10.4269/ajtmh.2002.67.162 . PMID 12389941.
- 1 2 3 González C (January 29, 2020). "Una bacteria presente en ácaros causa un raro tipo de infección en el sur". El Mercurio (in Spanish). p. A9.