Abstract
A fixed drug eruption (FDE) is a relatively common reaction associated with more than 100 medications. It is defined as a same-site recurrence with exposure to a particular medication. The primary approach and treatment for all types of FDEs are to identify and remove the causative agent, often accomplished by a thorough history of medication and other chemical exposures, and possibly prior episodes. The most common category of FDE, localized FDE, whether bullous or non-bullous, is self-limited. Although one can confirm the causative agent using oral challenge testing, it is not recommended due to the risk of severe exacerbation or possible generalization; patch testing is now preferred. Bullous FDE may resemble erythema multiforme. Treatment of localized FDE includes medication removal, patient counseling, and symptomatic relief. Failure to remove the causative agent in localized FDE can lead to recurrence, which is associated with increased inflammation, hyperpigmentation, and risk of a potentially lethal generalized bullous FDE (GBFDE), which may resemble Stevens–Johnson syndrome (SJS) or toxic epidermal necrolysis (TEN). Distinguishing GBFDE from SJS and TEN is salient and will be stressed: GBFDE has more rapid onset in 1–24 h rather than in weeks, less or no mucosal involvement, less or no systemic involvement, and a tendency for a more favorable prognosis; however, recent experience suggests it may be just as life-threatening. This review will provide a comprehensive update and approach to diagnosis and management.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
A fixed drug eruption (FDE) is a common drug eruption with distinctive morphology and etiology that ranges from antibiotics to cryptic exposure such as to quinine in a gin and tonic. |
Generalized bullous FDE may resemble SJS/TEN and may prove to be as deadly. |
1 Introduction
A fixed drug eruption (FDE) is often a clinically striking allergic phenomenon characterized by an erythematous or violaceous circular patch, plaque, or bullae with a somewhat dusky-grey center denoted for same-site recurrence after re-exposure to a specific drug, often with more involved sites with each recurrence [1,2,3]. FDE is a delayed type IV hypersensitivity reaction [4, 5]. First described by Bourns [6] in England in 1889, the term FDE was coined by the Frenchman Brocq [7] as an “éruption érythémato-pigmentée fixe” in 1894 to describe a reaction to antipyrine [7, 8]. Categorization of FDE is based on its clinical morphology, the major types being localized pigmenting, localized bullous, mucosal, non-pigmenting, generalized, and generalized bullous. The localized pigmenting type of FDE is the most common type and is a self-limited reaction [9, 10]. Recognition and management of the rare generalized bullous FDE (GBFDE) is a clinical conundrum that may have life-threatening consequences. Lyell [10] has proclaimed that his original description of four patients with toxic epidermal necrolysis (TEN) contains two patients who actually had GBFDE. Difficulty in distinguishing GBFDE from TEN has long been a problem [3]. We concur that GBFDE is quite unusual, has no precise incidence figures available, and is often misdiagnosed and rarely documented.
2 Epidemiology
Drug-induced skin reactions occur in 2–5% of hospitalized patients and 1% of outpatients [11]. FDE is a relatively common cutaneous drug reaction, representing up to 14–22% of patients, second only to morbilliform reactions [4, 9, 12, 13]. In one survey, GBFDE affected mainly older patients (median age 78 years, range 68–84 years) [3], in whom this diagnosis and other special types of FDE may be missed [14]. In one small Indian survey, FDE was most common among patients aged 21–30 years and affected both sexes equally, the most common offenders being antimicrobials and non-steroidal anti-inflammatory drugs (NSAIDs), with fluoroquinolones and nitroimidazole most closely linked [15]. In a study of 450 Pakistani FDE patients, 69% of patients had the condition for at least 1 year before diagnosis [16]. Orally administered drugs are the most common cause of an FDE [2], with topical and intravaginal medications being less likely [17].
An FDE can occur after exposure to ultraviolet in the A or B range (UVA/UVB) or to food instead of a drug [8]. Sometimes referred to as a fixed food eruption, it is probably best viewed as a type of FDE. The distinction can be challenging, as exemplified by quinine, an antimalarial compound found in tonic water; quinine can produce FDE in people drinking tonic water by itself or in a gin and tonic [44]. Other foods, including kiwifruit, peanuts, cashew nuts, and asparagus, may rarely produce an FDE; however, sometimes foods may contain antibiotics or other drugs that can cause an FDE.
3 Pathogenesis
The immunological reaction at the FDE site has been extensively studied, with its pathological changes being well described [9]. During the first 8 h, the basal layer of the epidermis is damaged by exposure to the offending agent [2]. Resident epidermal memory CD8+ T-cell activation by medication antigens plays a key role as these CD8+ T cells release early cytotoxic mediators such as interferon-γ [18,19,20]. In the later stages, recruited CD8+ T cells, CD4+ T cells, and neutrophils cause damage to melanocytes and keratinocytes [19]. Damage to the melanocytes leads to melanin leakage into the dermis [9]. This injury is controlled by the activation of FoxP3+ regulatory T cells within 24 h, which abate the activation mediated by both central memory and resident memory CD8+ T cells [19, 21]. Patients who develop an FDE may have reduced amounts of FoxP3+ regulatory T cells in the lesional periphery [21, 22].
After discontinuation of the drug, the basal layer of the epidermis starts to regenerate and the inflammatory cells begin undergoing apoptosis [2]. During regeneration, the dermal macrophages undergo phagocytosis of the leaked melanin and remain at the site, leading to the observed residual hyperpigmentation [9]. The basal layer keratinocytes release interleukin (IL)-15 during regeneration, which leads to the formation of resident memory CD8+ T cells [23, 24]. The resident memory CD8+ T cells are implicated in the same-site recurrence of FDE [23, 25]. Resident memory CD8+ T cells have shown swift reactivation and release of interferon-γ upon drug provocation in vivo [21]. In addition, CD3+ CD8+ T cells with markers similar to resident memory CD8+ T cells are documented in the epidermis of latent FDE lesions [21]. These resident memory T cells have been implicated in other recurrent same-site conditions, including psoriasis and cutaneous T-cell lymphoma [24].
4 Genetics
Multiple types of drug hypersensitivity reactions, including FDE, have been associated with specific human leukocyte antigens (HLAs) [26]. These HLA genes produce major histocompatibility complex (MHC) molecules. In FDE, CD8+ T cells play a critical inflammatory role by recognizing certain drugs in association with specific MHC class I molecules found on keratinocytes [27]. MHC class I molecules are derived from HLA-A, HLA-B, and HLA-C genes in humans. There are several examples of HLA-A or HLA-B associated with FDE, including HLA-B22 with feprazone-induced FDE, and HLA-A30 with trimethoprim-sulfamethoxazole-induced FDE [22, 27].
5 Clinical Presentation
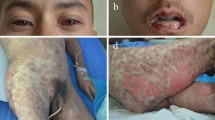
FDE is first evident as a red or violaceous circular plaque with a dusky-grey center [28]. The eruption may be solitary, a localized cluster, or diffuse. In one study, 16% of patients had a single plaque, 36% had two to five plaques, and 47% had more than five plaques on initial presentation [16]. FDE can appear at any skin site but targets areas with thin skin, such as the lip mucosa, genitals, and perianal sites [22]. Areas of prior cutaneous trauma such as burns, bites and healed herpes simplex virus (HSV) infection are favored targets [21]. FDEs occur up to 1 week after the first drug exposure, and, for subsequent exposures, between 30 min and 8 h afterwards [29]. FDE is usually asymptomatic, but may be painful or pruritic [30]. It usually resolves a few weeks after stopping the medication, but the post-inflammatory hyperpigmentation can last up to a few months afterwards (Fig. 1) [28]. GBFDE tends to be first evident as widespread well-demarcated, dusky-red or heavily pigmented oval patches, some polycyclic, with non-tense blisters and erosions. Typical clinical features that may aid in differentiating GBFDE from Stevens–Johnson syndrome/TEN are as follows: (1) blistering usually affects only a small percentage of body surface area, and between the large blisters there are sizable areas of intact skin; (2) erosive mucosal involvement is rare and, when evident, rather mild; (3) patients usually do not feel sick or have fever, and are generally in much better overall health than those with SJS/TEN; (4) most patients report a history of a similar, often local reaction.
5.1 Histology
FDE demonstrates changes in the epidermis and upper dermis [8, 31]. Histological changes include a vacuolar interface dermatitis, hydropic degeneration of the basal epidermal layer, spongiosis, a normal keratin layer, scattered individual keratinocyte necrosis, a perivascular lymphocytic and eosinophilic infiltrate, and pigmentary incontinence [31, 32]. FDE has overlapping pathologic features with erythema multiforme, SJS, and TEN, although FDE tends to show increased inflammation and many more dermal melanophages, as will be detailed shortly in discussing GBFDE.
5.2 Common Medications
Over 100 medications can cause FDE, with many common substances being implicated (Tables 1, 2) [33]. FDE is most commonly seen with trimethoprim-sulfamethoxazole, nitroimidazoles such as metronidazole and tinidazole, fluoroquinolones such as levofloxacin and ciprofloxacin, and NSAIDs [28, 33]. Other causative antibiotics include tetracyclines, amoxicillin, and rifampin [1, 30, 34]. Other analgesics include acetaminophen, salicylates, piroxicam, and mefenamic acid [1, 9]. Oral contraceptives, phenolphthalein-containing laxatives, barbiturates, and other sulfonamides are frequently involved [1, 33, 35]. In Singapore, NSAIDs caused half of the cases, with etoricoxib alone the most implicated [36]. Substances such as tartrazine in food additives and cold medication and quinine in alcoholic beverages, such as gin and tonic, may be overlooked, but should be considered [9]. Influenza vaccine may rarely be etiologic [28].
There are cases of cross-reactivity between drugs of similar structure [2]. Tetracyclines and trimethoprim-sulfamethoxazole most commonly show cross-sensitivity [1, 36]. Awareness of cross-reactivity is important in FDE as patients should be made aware of which medications may potentiate a recurrence [37].
6 Diagnosis
It is imperative to review the patient’s medications and history of consumed drugs [2]. Ingested drugs that are over-the-counter, herbal, and only occasionally taken should be included. The etiology can be difficult to elucidate in patients with multiple medications and a first-time episode [37].
FDE can sometimes be confirmed using oral challenge or patch testing [29]. Oral challenge has been the gold standard of diagnosis but is now usually contraindicated due to the risk of widespread FDE or GBFDE [5, 38,39,40]. The oral challenge test involves orally administering subtherapeutic doses of the suspected drug and assessing for a reaction [2]. One-tenth of the dose, at least 2 weeks after the last recurrence, has been the standard recommendation [9]. In a large study of 450 Pakistani cases, common adverse effects associated with the oral challenge test included pruritus, fever, and generalized urticaria [16].
Patch testing is the best confirmation method and is methodologically easier and safer than oral challenge [2]. Patch testing is conducted on a hyperpigmented site in an area of previous FDE, utilizing normal skin as a control. However, the sensitivity of patch testing depends on the reactivity of resident memory CD8+ T cells and drug permeability through the skin [37]. Patch testing is ineffective in patients without visible lesions as it relies on the resident memory CD8+ T cells that lie primarily in the hyperpigmented epidermis. Patch testing is a sensitive marker for NSAIDs, but not for antibiotics [2], and is therefore not considered a reliable testing method [37]. Patch testing should be performed a few weeks after the lesions resolve to avoid a false negative result due to a refractory period.
An uncommon method of FDE confirmation is performed using the lymphocyte transformation test (LTT), which aims to measure a sensitized T-cell reaction in response to the in vitro addition of the drug [41]. LTT is primarily used in confirming drug hypersensitivity reactions, where it has a sensitivity of 60–70%. LTT has not been shown to be an effective confirmatory test for FDE; however, selected reports have shown potential for LTT, as it has been diagnostic in specific cases of FDE caused by allopurinol or fluconazole [41, 42].
Biopsy is indicated in patients with an unclear diagnosis or associated systemic symptoms such as fever, malaise, or arthralgias, and is also indicated in the special subtypes of generalized FDE, GBFDE, and mucosal FDE.
6.1 Differential Diagnosis
FDE resembles erythema multiforme, both clinically and histopathologically [2]; however, FDE tends to be more localized, appears as oval-shaped patches, and does not contain the characteristic edematous halo associated with erythema multiforme or target lesions. Histopathologically, erythema multiforme, SJS, TEN, and FDE share similar epidermal changes; however, localized pigmenting FDE shows greater psoriasiform epidermal hyperplasia, more spongiosis, and prominent melanophage involvement [43]. Localized contact dermatitis, phytophodermatitis, and lichen planus can also resemble erythematous plaques of FDE [2, 45]. HSV can be mistaken for FDE, especially when mucosal membranes are involved or a reaction occurs at the site of a prior HSV lesion.
7 Special Subtypes
Mucosal FDEs target the oral and genital mucosa. Unlike classical FDE, mucosal FDE commonly has bullous or erosive lesions without residual hyperpigmentation [38]. Mucosal FDEs usually do have associated cutaneous findings. Oral mucosal lesions may occur on the tongue and hard palate. Naproxen and trimethoprim-sulfamethoxazole are the most commonly implicated medications [2]. The differential of isolated oral FDE includes herpes simplex labialis, pemphigus vulgaris, aphthous stomatitis, and Behçet’s disease [27, 28]. Male genital FDE most commonly affects the glans penis, likely caused by tetracyclines or trimethoprim-sulfamethoxazole [35, 36], while the vulva is affected in female patients, with NSAIDs being the commonly linked [2].
The non-pigmenting FDE (NPFDE) is a rare subtype, typically caused by pseudoephedrine [36, 40]. NPFDE presents similarly to localized pigmenting FDE; however, NPFDE does not have residual hyperpigmentation after resolution due to the lack of hydropic degeneration and pigment incontinence [40]. NPFDE also tends to have more symmetrical, well-circumscribed lesions than localized pigmenting FDE.
Generalized FDE is similar to localized pigmenting FDE but presents with multiple bilateral cutaneous macules. Generalized FDE spares the mucosa and typically occurs on the trunk and extremities.
7.1 Generalized Bullous Fixed Drug Eruption
Recurrent FDE is associated with an increased amount of inflammation and hyperpigmentation with each occurrence [33, 46, 47]. Recurrence increases the risk for GBFDE, a rare reaction with cutaneous bullae and vesicles (Fig. 2) [5, 33]. Despite the generally accepted idea that GBFDE has little, if any, systemic involvement and a substantially better prognosis than SJS or TEN, GBFDE is still potentially life-threatening, as documented in a recent study with a surprisingly high 22% mortality rate among elderly patients (13 of 58 died) [3]. One tends to see well-demarcated red-brown patches with overlying bullae on a background of diffuse hyperpigmentation. This mortality rate was only slightly lower with GBFDE than for those with SJS/TEN despite GBFDE having much less or no mucosal and visceral involvement (Table 3). These two groups did not differ in other preselected criteria for severity. GBFDE is defined by the involvement of at least three of six separated body areas, including the head and neck, trunk, upper limbs, lower limbs, and genital area [8, 48].
Since most cases of GBFDE involve < 10% of the skin, it commonly resembles SJS [5]. GBFDE is suggested when a patient presents with a history of similar previous episodes, an onset of 30 min to 1 day after drug ingestion, and lack or minimal mucosal involvement [5, 33, 34]. SJS/TEN usually occurs 1–4 weeks after drug ingestion and almost always involves the mucous membranes; SJS/TEN patients may have more severe systemic symptoms such as fevers and chills without a history of prior similar episodes [5].
An evaluation of a skin biopsy specimen is the preferred confirmation method (Table 4). GBFDE may also clinically resemble staphylococcal scalded skin syndrome, pseudoporphyria, and vancomycin-induced linear immunoglobulin (Ig) A bullous dermatosis [48,49,50,51,52,53,54], but these disorders are separable histologically. GBFDE shows increased inflammation with eosinophils, fewer necrotic keratinocytes, and more melanin-containing dermal macrophages compared with SJS/TEN [8, 48]. Nevertheless, GBFDE may have full-thickness epidermal necrosis, which histologically strongly resembles and may be almost indistinguishable from SJS and TEN [3, 33, 34, 47]. In that case, potentially valuable distinctions may be serum granulysin levels and the number of intraepidermal granulysin-expressing cells, which were found to be much lower than in SJS/TEN [8]. Granulysin, a lipid-binding protein, induces apoptosis in a mechanism involving caspases and other pathways, and may be elevated sera from patients with SJS/TEN [55]. Rapid immunochromatographic testing for serum granulysin may be useful for the prediction of SJS and TEN. Furthermore, GBFDE is associated with more dermal CD4-positive cells, including Foxp31 regulatory T cells, and fewer intraepidermal CD56-positive cells.
8 Treatment
The primary treatment of FDE is removal of the causative drug, the identification of which can at times be challenging [56]. Patients should be counseled to avoid the drug and be offered a list of cross-reacting medications. Medical management is symptomatic to relieve pruritus or pain [11]. For cutaneous pruritus, medium or high-dose topical corticosteroids or oral H1 antihistamines may be employed [10]. Mild oral lesions can usually be managed with viscous lidocaine, topical corticosteroids, or calcium carbonate. For those with severe oral involvement, hospitalization and feeding support may be required. In patients with erosive genital FDE, zinc oxide paste may be provided to reduce irritation and the risk of infection.
The primary treatment for generalized and GBFDE is removal of the causative drug, along with supportive care. Moderate-dose oral corticosteroids have not been shown to be effective but are often administered [49]. Identification of GBFDE affects the level of patient management, with those mislabeled as SJS/TEN more likely to receive a higher level of care [33]. Use of cyclosporine has been advocated based on favorable responses in a few patients [57, 58]. Relative to SJS/TEN, GBFDE has been considered to have a better prognosis and has the potential to be managed without hospitalization in mild cases [33]. However, with mortality at 22% among elderly patients in one series [5], we believe that intensive care and possible admission to a burns unit should be considered in severe cases.
References
Ben Fadhel N, Chaabane A, Ammar H, Ben Romdhane H, Soua Y, Chadli Z, et al. Clinical features, culprit drugs, and allergology workup in 41 cases of fixed drug eruption. Contact Dermat. 2019;81(5):336–40.
Pretzlaff KM, Pandya AG, Dominguez AR. Fixed Drug Eruptions. In: Hall JC, Hall BJ, editors. Cutaneous drug eruptions. London: Springer; 2015. p. 181–92.
Lipowicz S, Sekula P, Ingen-Housz-Oro S, et al. Prognosis of generalized bullous fixed drug eruption: comparison with Stevens–Johnson syndrome and toxic epidermal necrolysis. Br J Dermatol. 2013;168(4):726–32.
Zaouak A, Ben Salem F, Ben Jannet S, Hammami H, Fenniche S. Bullous fixed drug eruption: a potential diagnostic pitfall: a study of 18 cases. Therapie. 2019;74(5):527–30.
Mitre V, Applebaum DS, Albahrani Y, Hsu S. Generalized bullous fixed drug eruption imitating toxic epidermal necrolysis: a case report and literature review. Dermatol Online J. 2017;23(7):1–4.
Bourns D. Unusual effects of antipyrine. Br Med J. 1889;2:818–20.
Brocq L. Erupo erythemato pigmentée fixé due to antipyrone. Ann Dermatol Syphyligr. 1894;5:308.
Cho Y-T, Lin J-W, Chen Y-C, et al. Generalized bullous fixed drug eruption is distinct from Stevens–Johnson syndrome/toxic epidermal necrolysis by immunohistopathological features. J Am Acad Dermatol. 2014;70(3):539–48.
Flowers H, Brodell R, Brents M, Wyatt JP. Fixed drug eruptions: presentation, diagnosis, and management. South Med J. 2014;107(11):724–7.
Lyell A. Requiem for toxic epidermal necrolysis. Br J Dermatol. 1990;122:837–8.
Patell RD, Dosi RV, Shah PC, Joshi HS. Widespread bullous fixed drug eruption. BMJ Case Rep. 2014;2014:2013200584.
Khaled A, Kharfi M, Ben HM, et al. Cutaneous adverse drug reactions in children. A series of 90 cases. La Tunisie Medicale. 2012;90(1):45–50.
Sharma VK, Dhar S. Clinical pattern of cutaneous drug eruption among children and adolescents in North India. Pediatr Dermatol. 1995;12(2):178–83.
Shelley WB, Shelley ED. Consultations in dermatology: studies of orphan and unique patients. Cambridge: Cambridge University Press; 2006.
Tripathy R, Pattnaik KP, Dehury S, Patro S, Mohanty P, Sahoo SS, Mohanty S. Cutaneous adverse drug reactions with fixed-dose combinations: special reference to self-medication and preventability. Indian J Pharmacol. 2018;50(4):192–6.
Mahboob A, Haroon TS. Drugs causing fixed eruptions: a study of 450 cases. Int J Dermatol. 1998;37(11):833–8.
Hermida MD, Consalvo L, Lapadula MM, et al. Bullous fixed drug eruption induced by intravaginal metronidazole ovules, with positive topical provocation test findings. Arch Dermatol. 2011;147:250–1.
Shiohara T, Mizukawa Y. Fixed drug eruption: a disease mediated by self-inflicted responses of intraepidermal T cells. Eur J Dermatol. 2007;17(3):201–8.
Shiohara T. Fixed drug eruption: pathogenesis and diagnostic tests. Curr Opin Allergy Clin Immunol. 2009;9(4):316–21.
Mizukawa Y, Yamazaki Y, Teraki Y, et al. Direct evidence for interferon-gamma production by effector-memory-type intraepidermal T cells residing at an effector site of immunopathology in fixed drug eruption. Am J Pathol. 2002;161(4):1337–47.
Shiohara T, Ushigome Y, Kano Y, Takahashi R. Crucial role of viral reactivation in the development of severe drug eruptions: a comprehensive review. Clin Rev Allergy Immunol. 2015;49(2):192–202.
Li H, Wiederkehr M, Rao BK, et al. Peculiar unilateral fixed drug eruption of the breast. Int J Dermatol. 2002;41(2):96–8.
Mizukawa Y, Yamazaki Y, Shiohara T. In vivo dynamics of intraepidermal CD8+ T cells and CD4+ T cells during the evolution of fixed drug eruption. Br J Dermatol. 2008;158(6):1230–8.
Hoetzenecker W, Nägeli M, Mehra E, et al. Adverse cutaneous drug eruptions: current understanding. Semin Immunopathol. 2016;38:75–86.
Nomura T, Kabashima K, Miyachi Y. The panoply of αβT cells in the skin. J Dermatol Sci. 2014;76(1):3–9.
Pirmohamed M. Genetic factors in the predisposition to drug-induced hypersensitivity reactions. AAPS J. 2006;8(1):E20–6.
Özkaya E. Fixed drug eruption: state of the art. Journal der Deutschen Dermatologischen Gesellschaft. 2008;6(3):181–8.
Byrd RC, Mournighan KJ, Baca-Atlas M, Helton MR, Sun NZ, Siegel MB. Generalized bullous fixed-drug eruption secondary to the influenza vaccine. JAAD Case Rep. 2018;4(9):953–5.
Ahmed AM, Pritchard S, Reichenberg J. A review of cutaneous drug eruptions. Clin Geriatr Med. 2013;29(2):527–45.
Hager JL, Mir MR, Hsu S. Fluoroquinolone-induced generalized fixed drug eruption. Dermatol Online J. 2009;15(12):8.
Weyers W, Metze D. Histopathology of drug eruptions–general criteria, common patterns, and differential diagnosis. Dermatol Pract Concept. 2011;1(1):33.
Mithari HS, Gole PV, Kharkar VD, Mahajan SA. Generalized bullous fixed drug eruption to fluconazole; with cross-reactivity to tinidazole. Indian J Dermatol. 2019;64(4):335–7.
Dharamsi FM, Michener MD, Dharamsi JW. Bullous fixed drug eruption masquerading as recurrent Stevens Johnson syndrome. J Emerg Med. 2015;48(5):551–4.
Sereflican B, Ozden HK, Goksugur N, Boran C, Parlak AH. Levofloxacin-induced generalized bullous fixed drug eruption. Dermatol Sin. 2016;34(3):164–5.
Sehgal VN, Srivastava G. Fixed drug eruption (FDE): changing scenario of incriminating drugs. Int J Dermatol. 2006;45(8):897–908.
Heng YK, Yew YW, Lim DS, Lim YL. An update of fixed drug eruptions in Singapore. J Eur Acad Dermatol Venereol. 2015;29:1539–44.
Andrade P, Brinca A, Gonçalo M. Patch testing in fixed drug eruptions: a 20-year review. Contact Dermat. 2011;65(4):195–201.
Özkaya E. Oral mucosal fixed drug eruption: characteristics and differential diagnosis. J Am Acad Dermatol. 2013;69(2):e51–8.
Bilgili S, Calka O, Karadag A, Akdeniz N, Kosem M. Nonsteroidal anti-inflammatory drugs-induced generalized fixed drug eruption: two cases. Hum Exp Toxicol. 2012;31(2):197–200.
Bellini V, Bianchi L, Hansel K, Finocchi R, Stingeni L. Bullous nonpigmenting multifocal fixed drug eruption due to pseudoephedrine in a combination drug: clinical and diagnostic observations. J Allergy Clin Immunol Pract. 2016;4(3):542–4.
Kim M-H, Shim E-J, Jung J-W, Sohn S-W, Kang H-R. A case of allopurinol-induced fixed drug eruption confirmed with a lymphocyte transformation test. Allergy Asthma Immunol Res. 2012;4(5):309.
Demir S, Cetin EA, Unal D, et al. Generalized fixed drug eruption induced by fluconazole without cross-reactivity to itraconazole: lymphocyte transformation test confirms the diagnosis. Drug Saf Case Rep. 2018;5(1):2.
Fung MA. Inflammatory diseases of the dermis and epidermis. Dermatopathology. Amsterdam: Elsevier Inc.; 2010.
Sohn KH, Kim BK, Kim JY, Song WJ, Kang HR, Park HW, et al. Fixed food eruption caused by Actinidia arguta (Hardy Kiwi): a case report and literature review. Allergy Asthma Immunol Res. 2017;9(2):182–4.
Janusz SC, Schwartz RA. Phytophotodermatitis: an occupational and recreational dermatosis in the limelight. Cutis (in press).
Shiohara T, Mizukawa Y. Fixed drug eruption: easily overlooked but needing new respect. Dermatology. 2002;205(2):103–4.
Croda L, Valentin M, Caufield M, Goldberg M, Pasieka H. A diagnostic dilemma: a case of generalized bullous fixed drug eruption versus Stevens–Johnson syndrome: 4249. J Am Acad Dermatol. 2017;76(6):AB13.
Lee C-H, Chen Y-C, Cho Y-T, Chang C-Y, Chu C-Y. Fixed-drug eruption: a retrospective study in a single referral center in northern Taiwan. Dermatol Sin. 2012;30(1):11–5.
Kauppinen K, Stubb S. Fixed eruptions: causative drugs and challenge tests. Br J Dermatol. 1985;112(5):575–8.
Bakke JR, Chantara A, Cash J, Fisher KR. Vancomycin-induced linear IgA bullous dermatosis presenting as generalized fixed drug eruption. J Cutan Pathol. 2019;46(12):979–81.
Schwartz RA, McDonough PH, Lee BW. Toxic epidermal necrolysis. Part I. Introduction, history, classification, clinical features, systemic manifestations, etiology, and immunopathogenesis. J Am Acad Dermatol. 2013;69:173–84.
Schwartz RA, McDonough PH, Lee BW. Toxic epidermal necrolysis. Part II. Prognosis, sequelae, diagnosis, differential diagnosis, prevention, and treatment. J Am Acad Dermatol. 2013;69:187–202.
Handler MZ, Schwartz RA. Staphlococcal scalded skin syndrome: diagnosis and management in children and adults. J Eur Acad Dermatol Venereol. 2014;28:1418–23.
Papadopoulos AJ, Schwartz RA, Fekete Z, Kihiczak G, Samady JA, Atkin SH, et al. Pseudoporphyria. An atypical variant resembling toxic epidermal necrolysis. J Cutan Med Surg. 2001;5:479–85.
Fujita Y, Yoshioka N, Abe R, Murata J, Hoshina D, Mae H, et al. Rapid immunochromatographic test for serum granulysin is useful for the prediction of Stevens–Johnson syndrome and toxic epidermal necrolysis. J Am Acad Dermatol. 2011;65(1):65–8.
Zawar V, Kirloskar M, Chuh A. Fixed drug eruption: a sexually inducible reaction? Int J STD AIDS. 2004;15:560–3.
Beniwal R, Gupta LK, Khare AK, Mittal A, Mehta S, Balai M. Cyclosporine in generalized bullous-fixed drug eruption. Indian J Dermatol. 2018;63(5):432–3.
Malviya N, Cyrus N, Vandergriff T, Mauskar M. Generalized bullous fixed drug eruption treated with cyclosporine. Dermatol Online J. 2017;23(2):1–3.
Acknowledgements
The authors thank their distinguished colleague, Prof. Dr. Vijay Zawar, Maharashtra University of Health Sciences, Maharashtra, India, for contributing Fig. 1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this work.
Conflict of interest
Shreya Patel, Ann M. John, Marc Zachary Handler, and Robert A. Schwartz have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Patel, S., John, A.M., Handler, M.Z. et al. Fixed Drug Eruptions: An Update, Emphasizing the Potentially Lethal Generalized Bullous Fixed Drug Eruption. Am J Clin Dermatol 21, 393–399 (2020). https://doi.org/10.1007/s40257-020-00505-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40257-020-00505-3