Practice Essentials
Leptospirosis is an infectious disease of humans and animals that is caused by pathogenic spirochetes of the genus Leptospira. It is considered the most common zoonosis in the world and is associated with settings of poor sanitation and agricultural occupations involving contact with animals or water. Increasingly, adventurous travel and "mud run" sports or races involving fresh water or soil exposure put humans at risk. Affected reservoir species experience chronic colonization of proximal renal tubules and shed leptospires in urine. Humans are not typically considered reservoirs but may chronically shed leptospires and may act as reservoirs in areas of high endemicity and poor waste sanitiation.
Leptospirosis is endemic in most areas where dengue virus is transmitted and may be mistaken for dengue, which is typically more common. Co-infection occurs in up to 8% of cases. Leptospirosis may also co-infect with dengue and other regionally prevalent pathogens, such as malaria, hantavirus, and scrub typhus. Leptospirosis is typically a biphasic pattern of early flu-like, septicemic illness followed by an inflammatory second phase. The latter may be characterized by systemic inflammatory response syndrome (SIRS) or cytokine storm. COVID-19 may follow a similar trajectory and may complicate diagnosis in the subtropics and tropics. These issues may complicate the diagnosis, especially in travelers or migrants presenting to non-endemic areas. It is important to consider leptospirosis when dengue is diagnosed in a severely ill patient, because early antibiotics are beneficial.
Furthermore, as climate change disrupts regional rainfall and temperatures, Leptospirosis should be considered in the setting of potential exposures. Molecular diagnostics and expanding access to these tools may remedy chronic under-detection and under-reporting in affected areas.
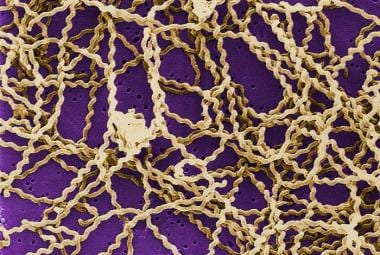 A scanning electron micrograph depicting Leptospira atop a 0.1-µm polycarbonate filter. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), Rob Weyant.
A scanning electron micrograph depicting Leptospira atop a 0.1-µm polycarbonate filter. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), Rob Weyant.
Signs and symptoms
Leptospirosis ranges in severity from no symptoms to a mild illness suggesting a viral infection to a multisystemic syndrome with unique features. It is characterized by sudden onset of the following:
-
Fever (38-40°C)
-
Rigors
-
Headache, retro-orbital pain, photophobia
-
Muscle pain localized to the calf and lumbar areas
-
Conjunctival suffusion
-
Dry cough
-
Nausea and vomiting, diarrhea
More severe disease manifests as icteric leptospirosis, also known as Weil disease, with the following features:
-
Icterus or frank jaundice
-
Renal failure with oliguria
-
Hemorrhagic features
-
Systemic inflammatory syndrome or shock
See Clinical Presentation for more detail.
Diagnosis
Laboratory studies used to screen for the diagnosis of leptospirosis include the following:
-
Leptospira immunoglobulin M (IgM) ELISA or IgM/immunoglobulin G (IgG) enzyme-linked immunoabsorbent assay (ELISA), including rapid diagnostic kits usable in the field
-
Real-time DNA polymerase chain reaction (PCR) of blood, urine, and cerebrospinal fluid (CSF)
Laboratory studies used to confirm the diagnosis of leptospirosis include the following:
-
Microscopic agglutination testing (MAT; criterion standard for serologic identification of leptospires, available at reference laboratories)
-
Single titer ≥1:200 or 4-fold rise in serum drawn between the first and fourth week of illness is considered diagnostic
-
DNA PCR of blood, urine, CSF, tissue
-
Culture of leptospires from body fluids or tissue (criterion standard, but requires specific media and several weeks’ incubation, thus usually limited to reference laboratory)
Studies to determine the extent of organ involvement and severity of complications may include the following, depending on the clinical presentation:
-
Complete blood cell (CBC) count
-
Renal function studies
-
Coagulation studies
-
Liver function studies
-
CSF analysis
-
Chest radiography
-
Biliary tract ultrasonography
-
Electrocardiography (ECG)
See Workup for more detail.
Management
Antibiotic therapy in the treatment of mild leptospirosis is typically unnecessary, since it is often self-limited, and most cases resolve without medical attention. Oral antibiotics shorten the course of illness and, perhaps most importantly, reduce and shorten urinary excretion of leptospires. Prophylaxis may be used in the setting of epidemics. If used, antibiotic treatment may include the following:
-
Doxycycline
-
Ampicillin or amoxicillin
-
Azithromycin or clarithromycin
-
Fluoroquinolone such as ciprofloxacin or levofloxacin
Antibiotics for leptospirosis requiring hospitalization include the following:
-
Intravenous penicillin G
-
Intravenous third-generation cephalosporins (cefotaxime and ceftriaxone)
-
Intravenous ampicillin or amoxicillin (second-line agents)
-
Intravenous erythromycin (in penicillin-allergic pregnant women)
Patients with severe cases of leptospirosis also require supportive therapy and careful management of renal, hepatic, hematologic, and central nervous system complications. If renal failure ensues, early initiation of hemodialysis or peritoneal dialysis may reduce mortality by nearly two-thirds. Additional supportive care may include inotropic agents, diuretics, or ophthalmic drops.
See Treatment and Medication for more detail.
Background
Leptospirosis is an infectious disease of humans and animals that is caused by pathogenic spirochetes of the genus Leptospira. It is considered the most common zoonosis in the world. [1] Leptospirosis is distributed worldwide (sparing the polar regions) but is most common in the tropics.
Leptospirosis has been recognized as a re-emerging infectious disease among animals and humans [2] and has the potential to become even more prevalent with advancing climate change and related trends. [3] By 2100, various climate models project that global temperatures will rise by 2°F to 11.5°F, depending on trends in future greenhouse gas emissions. [4]
Leptospira species infect a wide range of animals, including mammals, birds, amphibians, and reptiles. Part of its life cycle is a chronic carrier state in some animals, in which the leptospires spread hematogenously, colonize the proximal renal tubules, and are shed via urine into the environment. This may be fairly asymptomatic such that the animal becomes a chronic carrier and environmental reservoir. Humans can shed leptospires for only a limited time and are therefore considered accidental hosts. Rats, dogs, and ungulates, however, can chronically shed leptospires. The organism is typically transmitted via exposure of mucous membranes or abraded skin to the body fluid of an acutely infected animal or by exposure to soil or fresh water contaminated with the urine of a chronic carrier.
Occupational exposure probably accounts for 30-50% of human cases of leptospirosis. The main occupational groups at risk include farm workers, veterinarians, pet shop owners, field agricultural workers, abattoir workers, plumbers, meat handlers and slaughterhouse workers, coal miners, workers in the fishing industry, military troops, milkers, and sewer workers.
Although leptospirosis continues to be predominantly an occupational disease, in recent decades it has also increasingly been recognized as a disease of recreation. The disease may be acquired during "adventure racing," travel, or sporting events that involve fresh water or hiking (eg, "mud runs"). [5, 6, 7, 8, 9] Urban dwellers in economically deprived areas may contract the disease through exposure to rat urine. [10]
The incidence of leptospirosis is rising in domestic dogs, especially those that live on dairy farms or roam in rural areas. Leptospirosis in a pet increases the risk of human transmission if there is contact with the urine, feces, birth products, or tissues of the animal. Other routine types of contact and care pose little risk. More exotic pets such as rodents have transmitted leptospirosis to owners, and reptiles can also act as chronic carriers.
Widespread flooding may lead to epidemic spread of leptospirosis in large populations. [11] Hurricanes in subtropical and tropical areas pose a risk of leptospirosis outbreaks. Flooding on a smaller scale may also lead to individuals contracting the disease. [12]
In 90% of cases, leptospirosis manifests as an acute febrile illness with a biphasic course and an excellent prognosis. Nonspecific signs and symptoms of leptospirosis (eg, fever, headache, nausea, vomiting) are often confused with viral illness.
In 10% of cases, the presentation is more dramatic, and the infection has an overall case fatality rate of 5%-10%. Known as Weil disease or icteric leptospirosis, the classic definition of this form of leptospirosis includes fever, jaundice, renal failure, and hemorrhage. Other organ systems (ie, pulmonary system, cardiac system, central nervous system) are also frequently involved. Risk factors associated with higher mortality risk include old age, alteration of mental status, and pulmonary alveolar hemorrhage. (See Clinical Presentation.)
Treatment of leptospirosis should be started as soon as possible. Treatment is begun empirically in patients with a plausible exposure history and compatible symptoms, as culture times for Leptospira are long and recovery rates are low. The criterion standard for serologic identification of leptospires, microscopic agglutination testing (MAT), is available only at reference laboratories. Paired acute and convalescent serum specimens can provide delayed confirmation of the diagnosis.
In uncomplicated infections that do not require hospitalization, oral doxycycline has been shown to decrease duration of fever and most symptoms. In hospitalized patients, intravenous penicillin G has been the treatment of choice. Patients with severe leptospirosis (Weil disease) require supportive therapy and careful management of renal, hepatic, hematologic, and central nervous system complications. (See Treatment.)
Historical background
In ancient China, a disease that was certainly leptospirosis was recognized as an occupational hazard of rice harvesters. In Japan, leptospirosis was called akiyami, or autumn fever, a term still used for this disease. [13]
In the West, leptospirosis was described by Larrey in 1812 as fièvre jaune among Napoleon's troops at the siege of Cairo. The disease was initially believed to be related to the plague but not as contagious. Throughout the remainder of the 19th century, leptospirosis was known in Europe as bilious typhoid.
Leptospirosis was recognized as an occupational disease of sewer workers in 1883. In 1886, Adolph Weil published his historic paper describing the most severe form of leptospirosis that would be later known as Weil disease. Weil described the clinical manifestations in 4 men who had severe jaundice, fever, and hemorrhage with renal involvement. [14]
In 1907, Stimson used special staining techniques in the postmortem examination of a kidney from a person with Weil disease and found a spiral organism with hooked ends, which was named Spirochaeta interrogans because its shape resembled that of a question mark. Inada et al identified the causal agent of infectious jaundice in Japan in 1916, naming the organism Spirochaeta icterohaemorrhagiae. [14]
Pathophysiology
Leptospires are thin, coiled, gram-negative, aerobic organisms 6-20 µm in length. They are motile, with hooked ends and paired axial flagella (one on each end), enabling them to burrow into tissue. Motion is marked by continual spinning on the long axis. They are unique among the spirochetes in that they can be isolated on artificial media.
 A scanning electron micrograph depicting Leptospira atop a 0.1-µm polycarbonate filter. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), Rob Weyant.
A scanning electron micrograph depicting Leptospira atop a 0.1-µm polycarbonate filter. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), Rob Weyant.
Leptospires belong to the order Spirochaetales and the family Leptospiraceae. Traditionally, the organisms are classified based on antigenic differences in the lipopolysaccharide envelopes that surround the cell wall. Serologic detection of these differences, therefore, is based on identifying serovars within each species. Based on this system, the genus Leptospira contains two species: the pathogenic Leptospira interrogans, with at least 218 serovars; and the nonpathogenic, free-living, saprophytic Leptospira biflexa, which has at least 60 serovars.
Current studies that classify the organisms based on DNA relatedness identify at least 7 pathogenic species of leptospires. However, organisms that are identical serologically may be different genetically, and organisms with the same genetic makeup may differ serologically. Therefore, some authors feel that the traditional serologic system is the most useful from a diagnostic and epidemiologic standpoint.
Animal reservoirs
Most leptospiral serovars have a primary reservoir in wild mammals, which continually reinfect domestic populations. The organism affects at least 160 mammalian species and has been recovered from rats, swine, dogs, cats, raccoons, cattle, mongooses, and bandicoots. [15, 16] The most important reservoirs are rodents, and rats are the most common source worldwide. In the United States, important leptospiral sources include dogs, livestock, rodents, and wild animals.
Many serovars are associated with particular animals. For example, L pomona and L interrogans are seen in cattle and pigs; L grippotyphosa is seen in cattle, sheep, goats, and voles; L ballum and L icterohaemorrhagiae are associated with rats and mice; and L canicola is associated with dogs. Other important serotypes include L autumnalis, L hebdomidis, and L australis. Leptospiral species' and serogroups' host animals vary from region to region. Individual animals may carry several serovars.
Leptospirosis in animals is often subclinical. Leptospires may persist for long periods in the renal tubules of animals by establishing a symbiotic relationship with little or no evidence of disease or pathological changes in the kidney. As a result, animals that serve as reservoirs of host-adapted serovars can shed high concentrations of the organism in their urine without showing clinical evidence of disease.
This leptospiruria in animals often occurs for months after the initial infection. Leptospiruria also has been found to occur in healthy immunized dogs. Leptospiruria in humans is more transient, rarely lasting more than 60 days. Humans and nonadapted animals are incidental hosts. With rare exceptions, man represents a dead end in the chain of infection because person-to-person spread of the disease is rare.
Transmission and incubation
Urinary shedding of organisms from infected animals is the most important source of these bacterial pathogens. Contact with the organism via infected urine or urine-contaminated media results in human infection. Such media include contaminated water and food, as well as animal bedding, soil, mud, and aborted tissue. Under favorable conditions, leptospires can survive in fresh water for as many as 16 days and in soil for as many as 24 days. [17]
Leptospires are believed to enter the host through the following:
-
Abrasions in healthy skin
-
Animal and rodent bites
-
Sodden and waterlogged skin
-
Mucous membranes or conjunctiva
-
Lungs (after inhalation of aerosolized body fluid) [18]
-
The placenta during pregnancy
Virulent organisms in a susceptible host gain rapid access to the bloodstream through the lymphatics, resulting in leptospiremia and spread to all organs, but particularly the liver and kidney. The incubation period is usually 5-14 days but has been described from 72 hours to a month or more.
Pathologic effects
Although direct invasion of tissue may cause some pathologic effects, researchers note that the marked degree of multiorgan tissue injury appears inconsistent with the number of leptospires found on microscopic examination of tissue. Other mediators induced by the leptospire are the suspected causes of the disease's various manifestations. Research has suggested endotoxin, hemolysin, and lipase as possible sources of pathogenicity. However, the true mechanism of host tissue injury remains unclear and likely involves a complex set of interactions.
The most consistent pathologic finding in leptospirosis is vasculitis of capillaries, manifested by endothelial edema, necrosis, and lymphocytic infiltration. Capillary vasculitis is found in every affected organ system. The resulting loss of red blood cells and fluid through enlarged junctions and fenestrae, which cause secondary tissue injury, probably accounts for many of the clinical findings.
In the kidneys, leptospires migrate to the interstitium, renal tubules, and tubular lumen, causing interstitial nephritis and tubular necrosis. Capillary vasculitis is readily identified. Although the glomeruli are spared, the progression from normal renal function to decreased glomerular filtration rate to renal failure requiring dialysis can be rapid. Renal failure is usually due to tubular damage, but hypovolemia from dehydration and from altered capillary permeability can also contribute to renal failure.
Liver involvement is marked by centrilobular necrosis and Kupffer cell proliferation. Jaundice may occur as a result of hepatocellular dysfunction.
Pulmonary involvement is secondary to alveolar and interstitial vascular damage resulting in hemorrhage. This complication is considered to be the major cause of leptospirosis-associated death.
Cardiac lesions have been identified in postmortem examinations. In an autopsy series of fatal cases of leptospirosis in Mumbai, India in 2005, involvement of the cardiovascular system was found in 41 of 44 cases. Interstitial myocarditis was the predominant feature on histopathological examination. These authors suggested that leptospirosis be viewed as an infective systemic vasculitis. [19]
Hemorrhage, focal necrosis, and inflammatory infiltration have been documented within the adrenal gland. Although these complications do not appear clinically, some researchers speculate that adrenal insufficiency may mediate, in part, the final vascular collapse associated with fatal leptospirosis.
The skin is affected by epithelial vascular insult. Skeletal muscle involvement is secondary to edema, myofibril vacuolization, and vessel damage. Muscular microcirculation is impaired and capillary permeability is increased, with resultant fluid leakage and circulatory hypovolemia.
The damage to the vascular system as a whole can result in capillary leakage, hypovolemia, and shock. Patients with leptospirosis may develop disseminated intravascular coagulation (DIC), hemolytic uremic syndrome (HUS), or thrombotic thrombocytopenic purpura (TTP). Thrombocytopenia indicates severe disease and should raise suspicion for a risk of bleeding. [20, 21]
If the host survives the acute infection, septicemia and multiplication of the organism persist until the development of opsonizing immunoglobulin in the plasma, followed by rapid immune clearance. Although the systemic immune response may eliminate the organism from the body, it may also lead to a symptomatic inflammatory reaction that can produce secondary end-organ injury.
Despite clearance from the blood, leptospires may remain in immunologically privileged sites, including the renal tubules, brain, and aqueous humor of the eye, for weeks to months. Persistent leptospires in the eye occasionally lead to chronic or recurrent uveitis. In humans, leptospires in the renal tubules and resulting leptospiruria rarely persist longer than 60 days.
Etiology
Leptospirosis is caused by spiral bacteria that belong to the genus Leptospira, the family Leptospiraceae, and the order Spirochaetales. These spirochetes are finely coiled, thin, motile, obligate, slow-growing aerobes.
The nomenclature system used to organize leptospires has been revised, making review of the literature often confusing. The traditional system divided the genus into 2 species: the pathogenic Leptospira interrogans and the nonpathogenic Leptospira biflexa. These species were divided further into serogroups, serovars, and strains based on shared antigens. L interrogans included more than 250 serovars.
The current classification system is based on DNA homology and recognizes the heterogeneity of the classic leptospires, dividing L interrogans and L biflexa into 20 named species. [22] Within these species, leptospires are further grouped by serogroups, serovars, and strains on the basis of microscopic agglutination testing (MAT).
Serologic grouping may, however, cross DNA-based species boundaries, and both pathogenic and nonpathogenic serovars may be found within the same species. There exist hundreds of serovars, and although certain species (eg, L interrogans) have a classic association with Weil disease, knowledge of the species type does not necessarily help predict disease severity.
Particular serovars may be associated with specific clinical manifestations. For example, a characteristic pretibial erythematous rash is seen in patients with L autumnalis infection, and gastrointestinal symptoms predominate in patients infected with L grippotyphosa.
Nevertheless, any leptospiral serovar can lead to the signs and symptoms seen with this disease. For example, jaundice occurs in 83% of patients with L icterohaemorrhagiae infection and in 30% of patients infected with L pomona. Aseptic meningitis commonly occurs in those infected with L pomona or L canicola.
Transmission of leptospires to humans typically occurs by invasion across mucosal surfaces or nonintact skin. Infection may occur via direct contact with infected animals or their tissues or urine or through contact with contaminated water and soil.
Epidemiology
United States statistics
Leptospirosis, as a clinical entity, is underdiagnosed and underreported. From 1985-1994, the reported annual incidence ranged from 0.02-0.04 cases per 100,000 persons. In 1994, 38 leptospirosis cases were reported nationwide, and the Council of State and Territorial Epidemiologists recommended removing leptospirosis from the list of notifiable diseases. [23]
Because reliable diagnostic testing was not readily available and organized reporting had not resulted in implementation of methods to control the disease, many states stopped reporting leptospirosis. Nevertheless, numerous states, including Hawaii, continued to report. In 2013, leptospirosis was reinstated as a nationally notifiable disease.
In Hawaii, which reports the highest annual occurrence rate, 405 suspected cases of leptospirosis were reported between June 1998 and February 1999; 61 of those cases were confirmed. [24] Case numbers widely varied from island to island within the state. Incidence rates ranged from 2.3-40.2 cases per 100,000 persons, with the highest numbers of cases on Kauai and Hawaii.
These reported cases starkly contrast with the prevalence rates found under active surveillance. [25] Using active surveillance measures, Hawaiian researchers projected the state's true local incidence at approximately 128 cases per 100,000 persons. Major risk factors identified in Hawaii include the use of water catchment systems, wild pig hunting, and the presence of skin wounds.
An estimated 100-200 cases are identified annually in the United States, with about 50% of cases occurring in Hawaii. Endemic canine leptospirosis is becoming more common in the United States, and California has seen a re-emergence of disease since 2000. Over the past 20 years, epidemiology has begun to shift from primarily recreational water exposures to an increasing number of occupational exposures related to farm and agricultural activities. [26]
Although less dramatic and often unrecognized, people with no obvious risk factor have significantly higher background infection rates than reports often indicate. Approximately 30% of children in urban Detroit [27] and 16% of adults in Baltimore [28] demonstrated serologic evidence of past infection. The Detroit study also showed correlation between degree of rat infestation and seropositivity rates. This finding suggests rats are major, if not the most important, vectors for human leptospirosis in the mainland United States.
Leptospirosis has a seasonal incidence. Most cases occur during the rainy season in the tropics and during the late summer or early fall in Western countries, when the soil is moist and alkaline. The leptospires from infected animals survive best in fresh water, damp alkaline soil, vegetation, and mud with temperatures higher than 22°C.
International statistics
Leptospirosis is a ubiquitous disease found throughout the world. Specific serovars vary with locality. The incidence varies from sporadic in temperate zones to endemic in a few tropical countries. Up to 80% of individuals in tropical areas are estimated to have positive seroconversion rates, indicating either past or present infection. Although leptospirosis is generally associated with tropical countries and heavy rainfall, most cases actually occur in temperate climates, probably because of underreporting in some countries. In addition, regional and local risk factors for leptospirosis include poor sanitation and open sewer systems in urban areas. The intersection of population density and contact with domesticated and wild animals also plays a role.
High-risk areas include the Caribbean islands, Central and South America, Southeast Asia, and the Pacific islands. Frequently, the disease gains public attention when outbreaks occur in association with natural disasters, such as flooding in Nicaragua in 1995 [11] or among foreign travelers, as with extreme athletes competing in tropical rainforests.
Sex- and age-related differences in incidence
No evidence suggests that leptospirosis affects persons of various races, ages, or sexes differently. However, because occupational exposure constitutes a major risk for development of disease, a disproportionate number of working-aged males seem to be affected.
On the other hand, the rates may be different because some practitioners are more likely to look for and, hence, diagnose the disease in patients who have obvious risk factors. When population groups other than adult males are actively surveyed, their rates are higher than those previously reported.
In addition, outbreaks have been reported in which more than 40% of patients were younger than 15 years, a reversal of traditional prevalence rates. Potential explanations in such cases include childhood predilections to play with suspected vectors (eg, dogs) or indiscriminate contact with water. Active surveillance measures have detected leptospire antibodies in as many as 30% of children in some urban American populations.
Seasonal variation in incidence
Leptospirosis has a seasonal incidence. Most cases occur during the rainy season in the tropics and during the late summer or early fall in Western countries, as leptospires survive best in fresh water, damp alkaline soil, vegetation, and mud with temperatures higher than 22°C.
Seasonal outbreaks associated with changes in local water levels have been described. Flood conditions increase risk for exposure to the population at large, and drought causes leptospire concentrations to peak in isolated pools. [29, 30]
Seasonal variation also correlates with participation in activities that increase exposure to leptospires. For example, outbreaks have been reported in triathlon athletes and white-water rafters. [6, 7, 8, 9]
Prognosis
The mild form of leptospirosis is rarely fatal, and an estimated 90% of cases fall into this category. The mortality rate in severe leptospirosis averages approximately 10%, but has been described as ranging from 5-40%. Elderly and immunocompromised people are at the highest risk for mortality overall. Most deaths occur from renal failure, massive hemorrhage, or acute respiratory distress syndrome (ARDS).
The incidence of pulmonary involvement has increased over the past few years, affecting up to 70% patients. Pulmonary involvement has emerged as a serious cause of mortality, becoming the main cause of leptospirosis-associated death in some countries. [31, 32]
Leptospirosis occurring during pregnancy is ominous. In a review of 16 cases, spontaneous abortion was likely during the first 2 trimesters. [33] When disease occurred in the third trimester, a third of pregnancies ended in abortion or perinatal death.
In general, survivors of leptospirosis experience little long-term morbidity, regardless of disease severity. Hepatic and renal functions return to normal, despite severe dysfunction during acute illness, even among patients who required dialysis.
Approximately a third of patients with documented aseptic meningitis may continue to complain of periodic headaches of varying severities. Patients who have had leptospiral uveitis may experience persistent visual acuity loss (caused by lens pigmentation following anterior uveitis) and blurry vision (associated with keratic precipitates in the anterior chamber).
Patient Education
Women who are pregnant or who are trying to become pregnant should avoid exposure to leptospirosis or may consider avoiding pregnancy during periods of high-risk exposure. While there are little data on congenital infection in humans, leptospirosis has been documented to cause spontaneous abortion and stillbirth during the first and second trimesters in over 60% of cases, probably in patients with more severe disease. [33] Women who become ill during the last trimester of pregnancy and have had high-risk exposure should present promptly for treatment to prevent in utero infection. Newborns of ill mothers can also be treated. Leptospires may be shed in breastmilk for an unknown duration.
Travelers and participants in triathlons, "adventure racing" or other freshwater sports who may be hiking and may otherwise be exposed to fresh water, soil, mud, and vegetation are at higher risk, especially those older than 60 years or those who are immunosuppressed. These individuals in particular should seek information concerning the risk for leptospirosis (and other infectious diseases) at their destinations before they travel, and take appropriate precautions. [9]
Patients should be advised to do the following:
-
Avoid contact with environments potentially contaminated with animal urine, especially rodent-infested areas.
-
Avoid swallowing or inhaling water from lakes, rivers, or swamps while swimming.
-
Shower promptly after swimming in fresh water and treat any cuts or abrasions with topical antibacterial medication and bandages.
-
Avoid participation in adventure racing activities with any cuts or abrasion of the skin.
-
Wear protective clothing and shoes.
Individuals who own dogs in warm climates with high rainfall may discuss vaccination with their veterinarian. At-risk animals are those that frequent areas where there are cattle, swine, rodents, or wild animals or that may drink from lakes, rivers, or streams in rural areas. Vaccination is repeated yearly.
Several polyvalent vaccines are available for immunization of livestock. Leptospirosis is a significant cause of stillbirths and reduced productivity in agriculture.
-
Darkfield microscopy of leptospiral microscopic agglutination test. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), M Gatton.
-
A scanning electron micrograph depicting Leptospira atop a 0.1-µm polycarbonate filter. (This image is in the public domain and thus free of any copyright restrictions. Courtesy of the Centers for Disease Control and Prevention (CDC), Rob Weyant.
-
Silver stain, liver, fatal human leptospirosis. Courtesy of the Centers for Disease Control and Prevention (CDC), Dr Martin Hicklin.