-
PDF
- Split View
-
Views
-
Cite
Cite
Leslie T Cooper, Tal Hasin, Margaret Ryan, Third time’s a harm? Cardiac risk of SARS-CoV-2 mRNA booster vaccines in younger men, European Heart Journal, 2024;, ehae157, https://doi.org/10.1093/eurheartj/ehae157
Close - Share Icon Share
COVID-19 vaccine-related risk and prevention strategies.
This editorial refers to ‘Booster vaccination with SARS-CoV-2 mRNA vaccines and myocarditis in adolescents and young adults: a Nordic cohort study’, by A. Hviid et al., https://doi.org/10.1093/eurheartj/ehae056.
COVID-19 vaccines have been critically important in reducing morbidly and mortality from SARS-CoV-2 infections. Providing continued protection against evolving SARS-CoV-2 pathogens has required administration of repeated doses of COVID-19 vaccines. In this issue of the European Heart Journal, Anders Hviid and colleagues provide important information about potential risks associated with repeated booster doses of COVID-19 vaccines.1
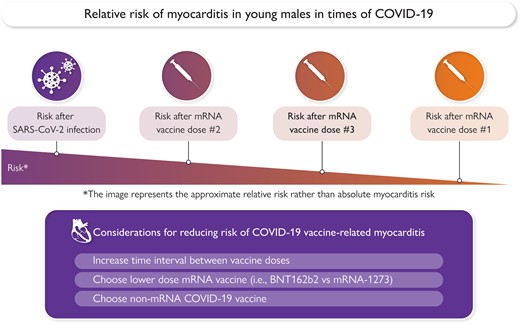
Vaccine-associated myocarditis gained widespread recognition after young men in the US military were reported to have an increased risk of myocarditis after the second dose of mRNA vaccine.2 Since the initial case series, multiple population-based reports have confirmed that post-vaccine myocarditis risk is greatest in young men between the ages of 12 and 39 years. The risk of myocarditis is also consistently higher after mRNA-1273 vaccine than after BNT162b2 vaccine, and when the time interval between vaccine doses is shorter.3–6 In the two-dose mRNA COVID-19 vaccine series, the risk of post-vaccine myocarditis is associated with the second dose of vaccine. In the demographic group of young males, risk after first vaccine dose is elevated above the background rate, but risk after the second vaccine dose may be 10- to 100-fold higher than the expected background rate.3–6
As COVID-19 recommendations expanded to include booster doses after the primary two-dose series, safety surveillance for myocarditis has been closely followed. Hviid and colleagues report that in the 28 days following a third dose of either BNT162b2 or mRNA-1273 vaccine, there was an increased rate of myocarditis in males between 12 and 39 years of age. Compared with the time window >28 days after the second dose, the relative risk was 2.08 for BNT162b2 and 8.89 for mRNA-1273. This translated to an absolute risk of 0.86/100 000 and 1.95/100 000 in males and 0.15/100 000 females in the 12–39 year age bracket. These data extend knowledge of third mRNA booster dose risk to a well-vaccinated population of 8.9 million individuals in Scandinavia.
The Scandinavian experience is very consistent with other global reports on risk of myocarditis after mRNA booster doses. Hviid and colleagues report briefly on the experiences of investigators in the USA, United Kingdom, France, Canada, Korea, and Israel. Investigators in Singapore, as well as those evaluating other data sources in the USA and Israel, have identified consistent associations with this safety signal.7–9 Overall, risk of myocarditis after the third dose of COVID-19 vaccine is lower than the risk after the second dose, but modestly higher than the risk after the first dose. Importantly, the risk of myocarditis after any dose of COVID-19 vaccine, in any population, is substantially lower than the risk of myocarditis after SARS-CoV-2 infection.
It is important to note that almost all evaluations, to date, address myocarditis risk after receipt of original formulations of mRNA COVID-19 vaccines. Understanding is limited for risk after additional boosters of the original vaccine formulation, subsequent bivalent formulation, omicron-specific vaccine formulations, or future COVID-19 vaccine formulations. One study in the USA, so far, has suggested that risk of myocarditis after fourth or fifth doses of COVID-19 vaccine is very low.10
The study by Hviid et al. has several limitations. The severity of myocarditis was not quantified. The clinical significance of the findings remains uncertain because many cases of post-vaccine myocarditis are relatively mild and resolve within a few weeks.11 It is also likely that patients who had myocarditis after the first or second vaccine dose did not get a booster dose. Similarly, only hospitalized patients were included in the cohort with myocarditis. Patients with mild disease or those who opted to not receive a third dose could be missed in this study design. The reliance on recorded diagnosis rather than magnetic resonance imaging- (MRI) or biopsy-based diagnosis limits the diagnostic certainty for myocarditis. Finally, because study enrolment ended in September of 2022, the most recent vaccines and the current circulating virus variants were not included in the evaluation.
Subclinical cases of myocarditis may be of importance both for the understanding of the biology and for possible long-term effects. In recent prospective investigations, a mild and transient increase in cardiac-specific troponin was observed days after vaccination in 0.6%–2.8% of the participants.12 Some caution is needed in attributing asymptomatic increases in troponin to myocarditis because the mechanism of injury is unknown and assay interference by macrotroponin complexes or heterophile antibodies can lead to false-positive troponin values.13 Interestingly, more women had post-vaccine myocardial injury in the prospective troponin analysis, in contrast to the male dominance demonstrated in Hviid’s work and other population-based studies. The interpretation of this discrepancy is yet to be delineated, together with multiple open questions on the implications of this subclinical phenomenon. However, the higher incidence of possible asymptomatic myocardial injury after COVID-19 mRNA vaccine suggests that clinical myocarditis cases may represent a subset of actual myocardial injury.
The mechanisms of cardiac injury after COVID-19 mRNA vaccines remain uncertain. Interleukin-1RA (IL-1RA) autoantibodies and hyperphosphorylated IL-1RA were higher in 14- to 21-year-old patients in whom myocarditis was confirmed by endomyocardial biopsy.14 In a second case series, full-length spike protein was detected in the plasma of individuals with post-vaccine myocarditis and was absent in vaccinated but asymptomatic controls.15
What are the implications of the accumulating data on myocarditis after COVID-19 mRNA vaccine? The treating physician who is now aware of this potential reaction can discuss this low risk against the potential benefits of vaccination on an individual basis. Individual risk may be assessed based on age, sex, type of vaccine, and the vaccine dose. An open question remains regarding individuals with prior myocarditis or post-vaccination chest pain. Cases of mildly symptomatic myocardial injury should be followed to determine whether mild injury has longer term clinical impacts. Policymakers should incorporate the information on adverse reactions into the decision regarding booster doses against new SARS-CoV2 strains, and future mRNA vaccines should be investigated in light of this potential reaction. As research continues, we hope that more information will be gained regarding the risk factors, biology, and long-term implications of this important topic.
Declarations
Disclosure of Interest
L.T.C. works as a consultant for Moderna Inc. regarding vaccine safety. All other authors declare no disclosure of interest for this contribution.
References
Author notes
The opinions expressed in this article are not necessarily those of the Editors of the European Heart Journal or of the European Society of Cardiology.