Answer
390.9k+ views
Hint: The Gay Lusaacc’s law of gaseous volume establishes a relationship between pressure and temperature of a fixed mass of gas kept at a constant volume. Knowing the basic aim of the Gay Lusaacc’s law helps in stating the law.
Complete step by step answer:
The Gay Lusaacc’s law of gaseous volume states that the pressure of a given mass of gas varies directly with the absolute temperature, when the volume is kept constant.
In simple terms we can explain that when gases react together they do so in volume which bears a simple whole number ratio provided that the temperature and the pressure of the reacting gases and their products remain constant.
Mathematically it can be written as,
P/T = k
Where, P is the pressure is the exerted by the gas
T is the absolute temperature of the gas
k is the constant
The relationship between the pressure and absolute temperature of a given mass of gas at constant volume can be illustrated graphically as follows:
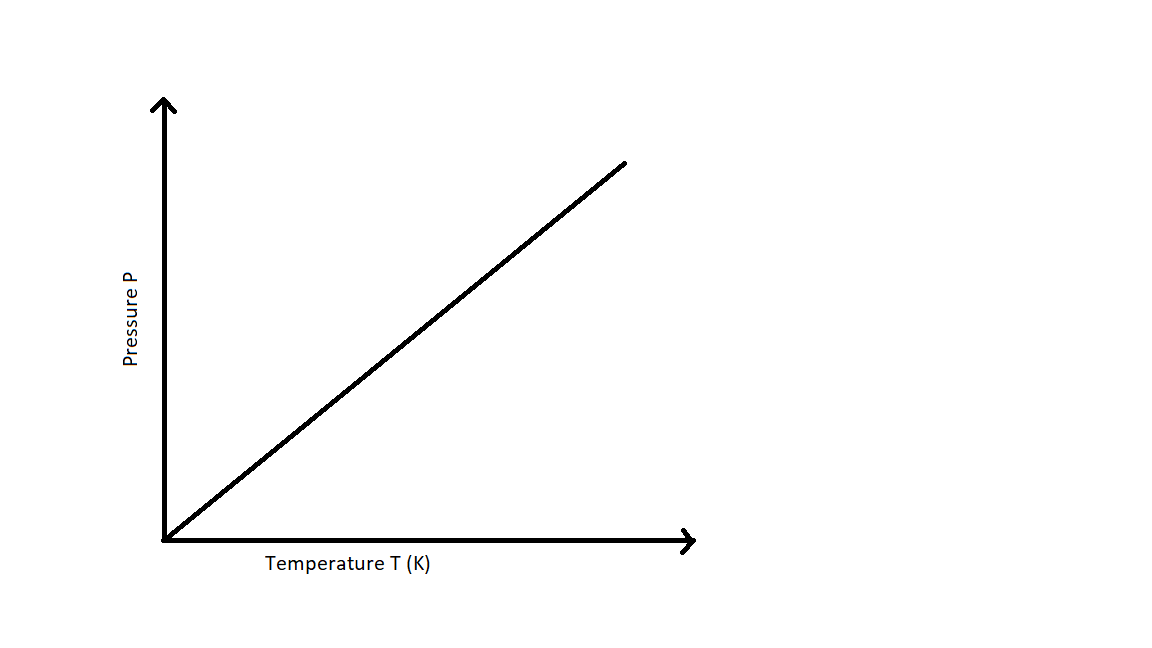
From the graph it can be understood that the pressure of the gas kept at constant volume reduces constantly as it is cooled until the gas eventually undergoes condensation and becomes a liquid.
Note: The Gay Lusaacc’s law can be explained with the help of an example such as in the chemical reaction of formation of hydrogen chloride gas, one volume of hydrogen is mixed with two volumes of chlorine. This results in the formation of two molecules of hydrogen chloride at constant temperature and pressure.
Complete step by step answer:
The Gay Lusaacc’s law of gaseous volume states that the pressure of a given mass of gas varies directly with the absolute temperature, when the volume is kept constant.
In simple terms we can explain that when gases react together they do so in volume which bears a simple whole number ratio provided that the temperature and the pressure of the reacting gases and their products remain constant.
Mathematically it can be written as,
P/T = k
Where, P is the pressure is the exerted by the gas
T is the absolute temperature of the gas
k is the constant
The relationship between the pressure and absolute temperature of a given mass of gas at constant volume can be illustrated graphically as follows:

From the graph it can be understood that the pressure of the gas kept at constant volume reduces constantly as it is cooled until the gas eventually undergoes condensation and becomes a liquid.
Note: The Gay Lusaacc’s law can be explained with the help of an example such as in the chemical reaction of formation of hydrogen chloride gas, one volume of hydrogen is mixed with two volumes of chlorine. This results in the formation of two molecules of hydrogen chloride at constant temperature and pressure.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

What is the stopping potential when the metal with class 12 physics JEE_Main

The momentum of a photon is 2 times 10 16gm cmsec Its class 12 physics JEE_Main

How do you arrange NH4 + BF3 H2O C2H2 in increasing class 11 chemistry CBSE

Is H mCT and q mCT the same thing If so which is more class 11 chemistry CBSE

Trending doubts
Difference Between Plant Cell and Animal Cell

Difference between Prokaryotic cell and Eukaryotic class 11 biology CBSE

What is BLO What is the full form of BLO class 8 social science CBSE

Change the following sentences into negative and interrogative class 10 english CBSE

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

The cell wall of prokaryotes are made up of a Cellulose class 9 biology CBSE

What organs are located on the left side of your body class 11 biology CBSE

Select the word that is correctly spelled a Twelveth class 10 english CBSE

a Tabulate the differences in the characteristics of class 12 chemistry CBSE



