Abstract
To evaluate the influence of early nutritional intake on the growth pattern of very preterm infants. This was an observational study including 109 newborns (< 32 weeks gestational age). Perinatal morbidities, nutritional therapy (first four weeks of life), and weight, length, and head circumference (HC) growth at term-equivalent age were evaluated. Growth restriction was defined as a difference > 1.2 SD between the birth and term age measurements. Growth restriction at term-equivalent age: 52.3% (weight), 42.9% (length), and 22% (HC). Morbidities were positively correlated with nutrition therapy and negatively correlated with the total energy provision: protein ratio. The duration of parenteral nutrition, the time to reach full enteral feedings, and the total energy provision: protein ratio were significantly correlated. Nutrient intake influenced weight, length, and HC growth, and cumulative energy deficit was significantly associated with HC growth restriction.
Conclusion: Perinatal morbidities interfere with nutritional therapy and early nutrient intake, leading to insufficient energy and energy provision: protein ratio for growth.
What is Known: • The intake of macronutrients early in life, mainly protein, is important for the optimal growth of pretem infants. • The severity of morbidities and low gestational ages impact the nutritional management of preterm infants. | |
What is New: • The number of morbidities, reflecting the severity of the neonatal clinical course, had a detrimental effect on the nutritional therapy and nutrients intake. • The inadequate energy provision per gram of protein ratio was significantly associated with growth restriction in all growth measures at the second week of life, persisting for head circumference up to the fourth week, highlighting the importance of its measurement, as it could be a precocious sign of development risk. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Postnatal growth restriction remains a great challenge in neonatal intensive care units (NICUs), particularly for very preterm newborn (NB) infants [1]. The factors influencing growth may vary, and the growth of these NBs is associated with cognitive and cerebral developmental outcomes [2]. In addition, the definition of postnatal growth restriction remains controversial, with different definitions proposed [3], and the main debate centers around whether it is best to use a cutoff level to classify the weight restriction at a predetermined time point or whether it would be preferable to consider the weight growth deficit between two time points [4,5,6,7,8,9,10]. Currently, there are two extrauterine growth restriction (EUGR) definitions for preterm NBs after birth: a cross-sectional definition, which uses a cutoff point at a predetermined age, and a longitudinal definition, which uses the difference between the measurements at two predetermined time points [4,5,6,7,8,9,10].
The postnatal growth pattern is the result of a sequence of physiological modifications. After early weight loss, a period of stable growth begins, with lean mass accretion and growth velocity rates similar to those of intrauterine growth [11]. Preterm NBs without severe morbidities can achieve growth rate trajectories equivalent to intrauterine patterns if they receive proper nutritional care according to the current protocols [11]. Recently, the European Society of Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) proposed recommendations for the nutritional management of preterm infants, emphasizing the interdependency of energy provided and macronutrient fractions [12], with an acceptable initial reduction of 7–10% in weight for preterm NBs that gradually reaches the birth percentile [13]. However, the ideal growth velocity of preterm NBs remains unclear [13].
Therefore, the identification of factors that impact the growth of preterm NBs may contribute to a better clinical understanding and nutritional approach, facilitating a reduction in the EUGR and improving neurodevelopment. The objective of this study was to evaluate the influence of perinatal morbidities and nutritional intake during the first four weeks of life on the growth of very preterm NBs.
Methods
Study design and population
This observational study was performed in a cohort of NBs included in the project “Analysis of Growth and Body Composition of Preterm Newborns with Bronchopulmonary Dysplasia”, approved by the Ethics Committee of Instituto Fernandes Figueira (IFF/FIOCRUZ), under opinion number CAAEE 50243615.0.0000.5269, in accordance with the Declaration of Helsinki. The parents/guardians of all included newborns signed the informed consent form before admission to the study.
The sample size and eligibility criteria were as follows: a convenience sample, including all NBs less than 32 weeks of gestational age (GA) born between December 2015 and April 2021 and admitted to the NICU of IFF/FIOCRUZ. Newborns with malformations at birth, congenital infections, genetic syndromes, fetal hydrops, inborn errors of metabolism, and death during hospitalization in the NICU were excluded.
Outcomes and morbidities
Exposure
The morbidities presented by the NBs, nutritional therapy, and nutrient intake received during the first four weeks of life.
Morbidities and risk factors
Sepsis (positive blood culture); intraventricular hemorrhage (IVH, at any stage, Papile's grading system) [14]; bronchopulmonary dysplasia (BPD, need for oxygen at 36 weeks corrected GA requiring any ventilatory assistance) [15]; retinopathy of prematurity (ROP) [16]; necrotizing enterocolitis (NEC, confirmed, stages II and III of Bell's classification) [17]; small for gestational age (SGA) at birth (birthweight < 10th percentile); need for invasive mechanical ventilation (IMV); and duration (days). For this study, we considered both the type and number of morbidities presented by each NB.
Nutritional factors
Time (days) elapsed from birth until the beginning of enteral feeds and to reach full enteral feedings (120 ml/kg/day); duration (days) of parenteral nutrition (PN); nutritional intake received during the first four weeks of life (amount of protein, lipid, and carbohydrate in g/kg/day).
Outcome
Growth measurements at term-equivalent age, as described in Milanesi et al. [18], using the Fenton growth chart as a reference [19]. For this study, the EUGR was defined as the difference in the Z score for the three anthropometric measures (weight, length, and head circumference - HC) greater than 1.2. SD between term-equivalent age and birth, based on Goldberg et al.’s criteria for malnutrition [20]. This difference in the Z score was evaluated individually for each anthropometric measure.
Nutritional protocol
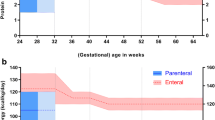
The nutritional therapy and protocol followed in this NICU were described in a previously published study [18]; in this unit, human milk use is widespread, with a multicomponent fortifier added when the diet volume reaches 100 ml/kg/day. Nutritional intake was registered prospectively during the first four weeks of life based on the nurses’ daily records using an electronic worksheet [21], as described in a previous study [18], which included the fluid volume (ml/kg/day), total energy provision (kcal/kg/day), protein (g/kg/day), lipids (g/kg/day), and carbohydrates (g/kg/day) received daily and the week’s mean.
In this study, the ratio of total energy provision (in kcal/kg/day) per gram of protein received was calculated by computing the energy provided by lipids and carbohydrates, and the ratio of 30 total energy provisions per gram of protein was considered the minimum necessary to provide growth [22]. Based on this ratio, the cumulative deficit in calorie: protein requirements for each week was calculated.
Statistical analysis
The data were analyzed with SPSS version 22.0 and R, version 4.0.3. Categorical variables are presented as absolute frequencies and percentages, and continuous variables are presented as the means or medians. The assumption of normality was verified with the Shapiro‒Wilk test. To identify the factors potentially associated with growth restriction for weight, length, and HC, a bivariate analysis was performed. For this study, we considered the presence of a morbidity as an independent categorical variable and the number of morbidities presented by each NB, if the NB presented one or more of the morbidities listed, as a discrete independent variable. The chi-square test, or the Fischer test when appropriate, was used to compare the categorical variables, and the Mann‒Whitney test was used to compare numerical variables between the groups with and without growth restriction. The Spearman correlation test was used to measure the degree of association between numerical variables. All analyses were performed considering a p value of 0.05.
Results
After applying the exclusion criteria, 109 newborns (GA < 32 weeks) were included, with a mean GA of 28.6 weeks (SD = 2.24). The mean maternal age was 26.9 years (SD = 6.9), with a mean schooling of 11.3 years (SD = 2.3); the mean paternal age was 30.2 years (SD = 7.0), with a mean schooling of 11.2 years (SD = 2.5).
Maternal hypertension was present in 30 (27.5%) mothers, and multiple gestations occurred in 64 pregnancies (58.7%) (Table 1). There was a similar distribution of sex among the NBs, and 12 NBs (11%) were born SGA (Table 1). Sepsis (21.1%), ROP (21.1%), and BPD (22.9%) occurred in approximately one-fifth of the patients, IVH in almost one-third (29.4%) and mechanical ventilation in almost half of the NBs (46.8%) (Table 1).
We had information on both measurements (birth and term-equivalent age) for weight in 101 NBs, for length in 91 NBs, and for HC in 92 NBs. The proportions of EUGR found were 52.3% (57/101), 42.9% (39/91), and 22% (24/92) for weight, length, and HC, respectively.
Being born SGA was associated with weight EUGR at term-equivalent age (p value: 0.011; chi-square test, Fischer correction) but not with length (p value: 0.195) or HC (p value: 0.722) growth deficits. In fact, of the 11 SGA NBs, 10 remained growth restricted for weight, 7 for length, and only 2 for HC. Mechanical ventilation contributed to the EUGR for both length (p value: 0.002; chi-square test) and HC (p value: 0.019; chi-square test) but not for weight (p value 0.841), while ROP (p value: 0.037; chi-square test) and BPD (p value: 0.043; chi-square test) were only associated with HC growth restriction; the other morbidities evaluated were not associated with growth restriction. When the morbidities were combined into two groups, with and without morbidities, the association was not significant (p value: 0.638 for weight restriction, 0.809 for length restriction, and 1.0 for HC growth restriction; chi-square test). A total energy provision: protein ratio less than 30:1 at the second week of life was significantly associated with weight EUGR (p value – 0.019, chi-square test) and length EUGR (p value – 0.004, chi-square test) but not HC EUGR (p value – 0.058, chi-square test).
The weight, length, and HC of the NBs that developed EUGR at term-equivalent age required significantly more time to begin enteral feeding, reach a full enteral feed volume, and remained on PN for a longer period (Table 2). The number of morbidities presented by the NB was significantly associated with length and HC EUGR but not with weight EUGR. Nutritional intake until the second week of life influenced weight, length, and HC growth, as indicated by the cumulative energy deficit at the second week (Table 2). However, for HC growth, inadequate nutritional intake during the first four weeks of life was reflected in cumulative energy deficit during this period that was significantly associated with EUGR (Table 2).
The number of morbidities had a weak correlation with the beginning of enteral feedings, but a significant and strong correlation was found with the duration of PN, the time to reach full enteral feeding, and the total energy provision: protein ratio in the second week. The duration of PN and the time to reach full enteral feeding were strongly correlated with the total energy provision: protein ratio in the second week (Fig. 1).
Correlogram of the number of morbidities, the number of days to begin enteral feeding, the number of days to reach full enteral feeding, the duration in days of parenteral nutrition, and the total energy provision per gram of protein during weeks 1, 2, 3, and 4 of life; the values were calculated with each week representing the mean of protein and calories. Legend: Positive and negative correlations are displayed in grayscales. The color intensity is proportional to the correlation coefficient, and correlations with a p value > 0.05 are considered nonsignificant. In this case, the correlation coefficients are left blank. N Morb: number of morbidities; PN: parenteral nutrition; Ratios 1, 2, 3, 4: total energy provision per gram of protein
Comparing the amounts of macronutrients administered during the first four weeks of life with those recommended by ESPGHAN [12], there was no significant difference in the amounts received by either group, with or without EUGR, and all NBs reached the recommended volume of fluids from the second week onward (Table 3). We evaluated whether the amounts of nutrients received by the NB, with or without EUGR, were within the range recommended by ESPGHAN. The EUGR NB received the minimum recommended protein amount by ESPGHAN at the fourth week and of carbohydrates at the second week; none of the EUGR NBs received the recommended amount of lipids during the first four weeks of life (Table 3). For the group without EUGR, the minimum amount of protein was attained at the third week, and the minimum amount of carbohydrate was attained at the second week; none of the groups reached the recommended amount of lipids (Table 3). None of the NBs, with or without EUGR, reached the recommendations for total calories per day during these first four weeks of life (Table 3).
Discussion
The incidence of EUGR at term-equivalent age found in this study, using a longitudinal definition and the Fenton growth chart (2013) [18], was 52.3% for weight, 42.9% for length, and 22% for HC, which is greater than those reported by González-García et al. [8] at discharge for weight (44.3%) and HC (13.7%), but not length (58.6%), using the same reference chart. Other studies reported a high incidence of weight EUGR using a longitudinal evaluation [4, 10]. With a transversal calculation and using the same reference chart at discharge [21], Tuzum et al. [7] and González-García et al. [8] reported an incidence of weight EUGR of 40.2% and 73.8%, respectively. The incidence of EUGR, either with longitudinal or transverse calculations, although varying among studies, remains high, which is a matter of concern.
In this study, we used the Fenton growth chart (2013) as a reference to calculate the Z scores [21]. The theoretical assumption behind the use of this chart was that when we compare the growth of preterm NBs with that of neonatal chart, we are considering the potential for growth of these preterm NBs, as if they were born at that time. Therefore, the influence of the interruption of intrauterine nutritional intake caused by premature birth, the energy cost of the morbidities faced by preterm NBs, and even the adaptative water dynamics of the first days of life are considered, when growth deficits are evaluated. Using the Fenton growth chart (2013), Rochow et al. demonstrated that preterm newborns without morbidities maintain a mean growth rate that is 0.8 SD lower and parallel to the intrauterine growth rate [11]. González-García et al. [8] and Yitayew et al. [23] compared Fenton (2013) [21] and Intergrowth 21 [24] charts to diagnose EUGR, and both studies reported a better definition of EUGR using longitudinal calculations; however, although there were some advantages of Intergrowth 21 over the Fenton (2013) chart, they did not affirm that one chart is superior to the other.
The ESPGHAN guidelines recommend the ideal intake of macro- and micronutrients to assure optimal growth for preterm infants with NB, stressing the importance of protein intake [12]. Lygerou et al. evaluated the effect of energy and macronutrients administered on preterm growth outcomes and reported that the total protein administered on Day 14 was a strong predictor of a positive increase in the weight Z score at discharge [25]. We analyzed the total energy provision: protein ratio and found that at the second week of life, a median ratio below 25 cal/g of protein was significantly associated with weight EUGR. This was even more evident concerning HC growth, and except for the third week, the total energy provision: protein ratios were significantly lower for those NB that remained restricted.
To improve lean mass accretion and, therefore, the optimal growth of preterm NBs, it is necessary to provide adequate amounts of protein and calories [13]. In the Embleton & van den Akker revision, the recommended supplement in enteral feedings is approximately 30–40 kcal/g of protein, and a lower intake of 25–30 kcal/g is sufficient if it is administered parenterally [20]. As shown in Table 3, this ratio increased during the first four weeks of life and reached the minimum requirement of 25 kcal/g of protein in NB without EUGR at the second week of life, except for HC growth. Notably, in our sample, the amount of protein received during the first four weeks of life was slightly less than that recommended by the ESPHGAN guidelines [12], and the amount of lipid received never reached the minimum recommended amount, with no difference between NBs with or without EUGR. Therefore, the inadequacy of this ratio was produced by the insufficient calories received, which is likely related to the amounts of lipids administered in relation to the protein received. In general, it is during the second week of life that the transition from PN to exclusive full enteral feedings occurs, and this period is considered critical for cumulative nutrient deficits and unsatisfactory growth, according to the ESPGHAN consensus [12]. In our sample, those NBs that developed EUGR remained in PN and reached full enteral feedings in a median of 13 days (weight and length EUGR) or 14 days (HC EUGR). The cumulative deficits in total energy provision per gram of protein found in this sample point to a greater challenge for these preterm NBs to overcome nutrient scarcity and attain their need for catch-up, not only for weight but also for length and HC.
The number of morbidities had a positive and significant correlation with the age at which the patient started an oral diet, the time elapsed to reach full enteral feedings, and the duration of PN. Additionally, there was a negative and significant correlation with the total energy provision: protein ratio in the first four weeks of life. Therefore, the severity of the clinical neonatal course had a direct and detrimental effect on nutritional intake during the first days of life, preventing the achievement of the ideal total energy provision: protein ratio and resulting in EUGR. This finding can partly explain why insufficient intake of nutrients by NBs is associated with a more severe clinical course. In critically ill NBs, who face life-threatening conditions, there is an acute metabolic stress response involving the activation of cytokines, hormones, and tissue catabolism, and the energy needs during the acute phases of disease are much greater in preterm NBs than in term NBs. As stated in Moltu et al.’s position paper of ESPGHAN for the nutritional management of critically ill newborns, this metabolic response involves three different phases: the early acute phase, late acute phase, and recovery phase [26]. Nutritional needs are different in each phase of the disease, and nutritional care should ideally be adapted to NB needs [26, 27]. However, critically ill NBs should receive nutritional support to cover their needs during the acute phases [26], and many of them receive the nutritional support needed by PN. Additionally, these preterm NBs face the energy demand of recovering from tissue damage. All these processes occur during the first or even second week of life, which corresponds to the worse intake restriction found in this sample. Even at the third and fourth weeks of life, the NBs still received the minimum total energy provision: protein ratio necessary for lean mass accretion. However, it is very challenging to balance the nutrients needed for these metabolic demands with the possible deleterious effect of excess nutrient administration, such as carbohydrates or nitrogen, which must be avoided.
The results of this study highlight the detrimental effects of inadequate intake on head growth, with a cumulative deficit of total energy provision for each gram of protein significantly greater in NBs that ended with HC EUGR at term-equivalent age. Poor head growth has been shown to be associated with impaired neurodevelopment [28]; total brain tissue and the intracranial cavity enlarges with increasing GA [29], and the free-fat mass Z score and fat mass Z score are associated with brain size Z scores in extremely preterm infants at term age [30]. Thus, as HC growth might signal brain growth, it is important to monitor its growth trajectory among preterm infants early in life, as the findings may indicate a worse development outcome.
One of the limitations of this study is the small sample size, which limited the ability to evaluate the associations between the morbidities presented and insufficient postnatal growth. Additionally, this small sample prevented the analysis of different strata of GA and the stratification of NBs that received mothers’ milk or not, which is currently being associated with cerebral development. Additionally, this study was performed in a single unit; therefore, the results cannot be generalized. This study considered only the longitudinal definition of growth restriction; therefore, we cannot extrapolate that the same results would be obtained considering the transversal definition of growth restriction. However, as this was a prospective study, all the data collected were reliable, with accurate measurements for the three anthropometric measures and precise calculations of nutrient intake, avoiding information bias.
Conclusion
This study, similar to others that found a great proportion of postnatal growth deficits, highlighted the challenge of nourishing preterm newborns, especially those who are critically ill. Early nutrient intake was affected by the presence of morbidities, and the second week of life appeared to be a critical period for predicting growth restriction for all three parameters: weight, length, and HC. Accordingly, it is important to monitor not only weight but also length and head circumference. More studies are necessary to ascertain the nutritional needs of critically ill preterm NB patients.
Data availability
The data presented in this study are available upon request from the corresponding author.
Abbreviations
- BPD:
-
Bronchopulmonary dysplasia
- ESPGHAN:
-
The European Society of Pediatric Gastroenterology, Hepatology and Nutrition
- EUGR:
-
Extrauterine growth restriction
- GA:
-
Gestational age
- HC:
-
Head circumference
- IFF/FIOCRUZ:
-
Instituto Fernandes Figueira
- IMV:
-
Invasive mechanical ventilation
- IVH:
-
Intraventricular hemorrhage
- NB:
-
Newborns
- NEC:
-
Necrotizing enterocolitis
- NICU:
-
Neonatal intensive care unit
- PMA:
-
Postmenstrual age
- PN:
-
Parenteral nutrition
- ROP:
-
Retinopathy of prematurity
- SGA:
-
Small for gestational age
References
Horbar JD, Ehrenkranz RA, Badger GJ, Edwards EM, Morrow KA, Soll RF et al (2015) Weight growth velocity and postnatal growth failure in infants 501 to 1500 Grams: 2000–2013. Pediatrics 136:e84–e92
Ong KK, Kennedy K, Castañeda-Gutiérrez E, Forsyth S, Godfrey KM, Koletzko B et al (2015) Postnatal growth in preterm infants and later health outcomes: a systematic review. Acta Paediatr 104:974–986
Fenton TR, Cormack B, Goldberg D, Nasser R, Alshaikh B, Eliasziw M et al (2020) Extrauterine growth restriction and postnatal growth failure are misnomers for preterm infants. J Perinatol 40:704–714
Renau MI, Aldecoa-Bilbao V, Esponera CB, de Mendoza BRH, Sanz MI, Iglesias-Platas I (2019) Applying methods for postnatal growth assessment in the clinical setting: evaluation in a longitudinal cohort of very preterm infants. Nutrients 11:2772
Reddy KV, Sharma D, Vardhelli V, Bashir T, Deshbotla SK, Murki S (2019) Comparison of fenton 2013 growth curves and intergrowth-21 growth standards to assess the incidence of intrauterine growth restriction and extrauterine growth restriction in preterm neonates ≤ 32 weeks. J Matern Fetal Neonatal Med 34(16):2634–2641
Maiocco G, Migliaretti G, Cresi F, Peila C, Deantoni S, Trapani B et al (2020) Evaluation of extrauterine head growth from 14–21 days to discharge with longitudinal intergrowth-21st charts: a new approach to identify very preterm infants at risk of long-term neurodevelopmental impairment. Front Pediatr 8:572930
Tuzun F, Yucesoy E, Baysal B, Kumral A, Duman N, Ozkan H (2018) Comparison of INTERGROWTH-21 and fenton growth standards to assess size at birth and extrauterine growth in very preterm infants. J Matern Fetal Neonatal Med 31:2252–2257
González-García L, García-López E, Fernández-Colomer B, Mantecón-Fernández L, Lareu-Vidal S, Suárez-Rodríguez M et al (2021) Extrauterine growth restriction in very low birth weight infants: concordance between fenton 2013 and INTERGROWTH-21st growth charts. Front Pediatr 9:690788
Viswanathan S, Osborn E, Jadcherla S (2022) Predictive ability of postnatal growth failure for adverse feeding-related outcomes in preterm infants: an exploratory study comparing Fenton with INTERGROWTH-21st preterm growth charts. J Matern Fetal Neonatal Med 35:5470–5477
Zozaya C, Díaz C (2018) Saenz De Pipaón M. How should we define postnatal growth restriction in preterm infants? Neonatology 114:177–180
Rochow N, Raja P, Liu K, Fenton T, Landau-Crangle E, Göttler S et al (2016) Physiological adjustment to postnatal growth trajectories in healthy preterm infants. Pediatr Res 79:870–879
Embleton ND, Moltu SJ, Lapillonne A, van den Akker CHP, Carnielli V, Fusch C et al (2023) Enteral nutrition in preterm infants (2022): a position paper from the ESPGHAN committee on nutrition and invited experts. JPGN 76:248–268
Embleton ND, Cleminson J, Zalewski S (2017) What growth should we aim for in preterm neonates? PCH 27:18–22
Papile LA, Burstein J, Burstein R, Koffler H (1978) Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 Gm. J Pediatr 92:529–534
Bancalari E, Jain D (2019) Bronchopulmonary dysplasia: 50 years after the original description. Neonatology 115:384–391
Chiang MF, Quinn GE, Fielder AR, Ostmo SR, Chan PRV, Berrocal A et al (2021) International classification of retinopathy of prematurity, 3rd ed. Ophthalmology 128:e51–e68
Bell MJ, Ternberg JL, Feigin RD, Keating JP, Marshall R, Barton L et al (1978) Neonatal necrotizing enterocolitis. Therapeutic decisions based upon clinical staging. Ann Surg 187(1):1–7
Milanesi BG, Lima PAT, Villela LD, Martins AS, Gomes-Junior SCS, Moreira MEL, Meio MDBB (2021) Assessment of early nutrition intake in preterm infants with bronchopulmonary dysplasia: a cohort study. Eur J Pediatr 180(5):1423–1430
Fenton TR, Kim JH (2013) A systematic review and meta-analysis to revise the fenton growth chart for preterm infants. BMC Pediatr 13:59
Goldberg DL, Becker PJ, Brigham K, Carlson S, Fleck L, Gollins L et al (2018) Identifying malnutrition in Preterm and neonatal populations: recommended indicators. J Acad Nutr Diet 118:1571–1582
Zin OA, Soares FVM, Abranches AD, Costa ACC, Villela LD, Moreira MEL (2019) Analysis of the differences between the prescribed and the administered diet to preterm infants using an electronic tool. Rev Paul Pediatr 37:472–478
Embleton ND, van den Akker CHP (2019) Protein intakes to optimize outcomes for preterm infants. Semin Perinatol 43:151154
Yitayew M, Chahin N, Rustom S, Thacker LR, Hendricks-Muñoz KD (2021) Fenton vs. intergrowth-21st: postnatal growth assessment and prediction of neurodevelopment in preterm infants. Nutrients 13:2841
Villar J, Giuliani F, Bhutta ZA, Bertino E, Ohuma EO, Ismail, et al (2015) Postnatal growth standards for preterm infants: the preterm postnatal follow-up study of the INTERGROWTH-21st project. Lancet Glob Health 3:e681–e691
Lygerou I, Ilia S, Briassoulis P, Manousaki A, Koropouli M, Hatzidaki E, Briassoulis G (2023) The impact of estimated energy and protein balances on extrauterine growth in preterm infants. Nutrients 15:3556. https://doi.org/10.3390/nu15163556
Moltu SJ, Bronsky J, Embleton N, Gerasimidis K, Indrio F, Köglmeier J et al (2021) Nutritional management of the critically ill neonate: a position paper of the ESPGHAN committee on nutrition. JPGN 73:274–289
Fivez T, Kerklaan D, Mesotten D, Verbruggen S, Wouters PJ, Vanhorebeek I et al (2016) Early versus late parenteral nutrition in critically ill children. N Engl J Med 374:1111–1122
Raghuram K, Yang J, Church PT, Cieslak Z, Synnes A, Mukerji A et al (2017) Head growth trajectory and neurodevelopmental outcomes in preterm neonates. Pediatrics 140:e20170216
Wang S, Fan P, Xiong D, Yang P, Zheng J, Zhao D (2018) Assessment of neonatal brain volume and growth at different postmenstrual ages by conventional MRI. Medicine 97:e11633
Binder C, Buchmayer J, Thajer A, Giordano V, Schmidbauer V, Harreiter K et al (2021) Association between fat-free mass and brain size in extremely preterm infants. Nutrients 13:4205
Funding
Partial financial support was received from Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro – FAPERJ – Grant number E-26/201.116/2021.
Author information
Authors and Affiliations
Contributions
Méio MDBB contributed to the conception of the study and was responsible for the coordination of the study, contributing to the analysis and interpretation of the data, the writing of the manuscript and its critical revision. Salgado GGM and Villela LD contributed to the analysis and interpretation of the data, the writing of the manuscript and its critical revision. Salgado GGM, Villela LD, Lima PAT, and Milanesi BG contributed to the material preparation and data collection. da Costa ACC performed the statistical analysis and contributed to the interpretation of the results and the writing of the manuscript. Moreira MEL contributed to the acquisition of financial resources for the study, the interpretation of the results and the critical revision of the manuscript. All the authors have read and approved the final manuscript and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Informed consent
Informed consent was obtained from all subjects involved in the study. This study was approved by the Ethics Committee of Instituto Fernandes Figueira (IFF/FIOCRUZ) on December 11th, 2015, under protocol code number CAAEE 50243615.0.0000.5269, in accordance with the Declaration of Helsinki.
Competing interests
The authors declare no competing interests.
Disclosure
All the authors state that artificial intelligence was not used during the entire process of interpreting the data, discussing the results, or writing the manuscript.
Additional information
Communicated by Daniele De Luca
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Méio, M.D.B.B., de Miranda Salgado, G.G., Villela, L.D. et al. Influence of morbidity, early nutritional intake, and total energy: protein ratio on longitudinal extrauterine growth restriction of very preterm newborns at term-equivalent age: an observational study. Eur J Pediatr (2024). https://doi.org/10.1007/s00431-024-05595-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00431-024-05595-3